| Jan 14, 2020 |
Artificial muscle sheets transform stem cells into bone
|
|
(Nanowerk News) Specifically programmed materials can, under specific conditions, encourage stem cells to transform into bone cells – as revealed by a German research team under the leadership of the Helmholtz-Zentrum Geesthacht, Centre for Materials and Coastal Research. To do this, the scientists implemented a so-called shape-memory polymer in stem cell research.
|
|
The study was published in Proceedings of the National Academy of Sciences of the USA ("Polymeric sheet actuators with programmable bioinstructivity").
|
|
Stem cells are known for their ability to turn into many different types of cell, be they muscle cells, cartilage, or bone cells. Just like the body they are part of, stem cells sense what happens around them and react accordingly. For decades, researchers have been learning how to steer this differentiation process by changing the cells’ environment. The knowledge acquired is already being used in tissue engineering, in other words, to generate substitute materials that restore or maintain damaged biological tissues.
|
|
However, most research has been done on static scaffolds. Now, researchers from the Helmholtz-Zentrum Geesthacht (HZG), the Berlin-Brandenburg Centre for Regenerative Therapies, the Freien Universität Berlin and the Helmholtz Virtual Institute for Multifunctional Biomaterials in medicine have used a dynamic scaffold.
|
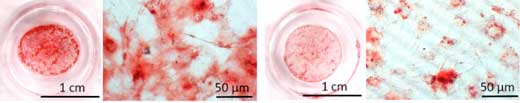 |
| The shape-memory polymer actuator sheet coated with stem cells shrunken when cold (left) and stretched when heated (right). (Image: Xu et al.) (click on image to enlarge)
|
New method created
|
|
The researchers took a polymer sheet that acts like an artificial muscle. The sheet has the unusual property in that it is trained to reversibly morph when exposed to repeated temperature changes. The researchers simply moulded a grid onto the underside of the sheet and programmed it to stretch as the temperature went from body temperature (37 °C) to 10 °C and to contract when re-heated. They then seeded the sheet with stem cells, and carefully observed the changing shape of the gridded sheet and cells.
|
|
With the help of this “artificial muscle”, the scientists could use one physical signal - the temperature change – to simultaneously send a second mechanical signal to the stem cells. With these synchronised stimuli it is possible to encourage the stem cells to turn themselves into bone cells.
|
|
“Our polymer actuator sheet has a so-called shape-memory function. In our experiments, this allows it to act like a transducer, with which we can effectively instruct the cells to do as we wish. We found that the changes in temperature, combined with the repeated stretching motion of the film was enough to encourage the stem cells to differentiate into bone cells” explained Professor Andreas Lendlein an author of the paper and head of the HZG’s Institute of Biomaterial Science in Teltow, Germany.
|
Potential application in complex bone fractures
|
|
“The programmed polymer sheets could, for example, later be used to treat bones broken so severely that the body can’t repair it by itself. Stem cells from a patient’s bone marrow could be cultured on the sheet and adaptively wrap around the bone during an operation. The previously “trained” cells could then directly strengthen the bones” said Professor Lendlein.
|
 |
| The stem cells grown on the polymer that changes shape at a programmed temperature could perhaps one day be used to support the healing of severely fractured bones. (Image: HZG/Xun Xu)
|
|
Given the recent report in New Scientist of a successful operation at 10 °C at the University of Maryland School of Medicine, such medical implants could become yet another tool in a surgeon’s toolkit.
|


