| Mar 13, 2023 |
A quick new way to screen virus proteins for antibiotic properties
|
|
(Nanowerk News) As conventional antibiotics continue to lose effectiveness against evolving pathogens, scientists are keen to employ the bacteria-killing techniques perfected by bacteriophages, the viruses that infect bacteria.
|
|
One major challenge standing in their way is the difficulty of studying individual bacteriophage (phage) proteins and determining precisely how the virus wields these tools to kill their host bacteria. New research from Lawrence Berkeley National Laboratory (Berkeley Lab) could help speed things along.
|
|
“We developed a high-throughput genetic screening approach that can identify the part of the bacterial cell targeted by a potent type of phage weapon called ‘single-gene lysis proteins,’” said Vivek Mutalik, a staff scientist in Berkeley Lab’s Biosciences Area and co-senior author on a new study describing the work in Nature Chemical Biology ("Multicopy suppressor screens reveal convergent evolution of single-gene lysis proteins"). “With rising antibiotic resistance, we urgently need antibiotic alternatives. Some of the smallest phages that we know of code for single-gene lysis proteins (Sgls), also known as ‘protein antibiotics,’ to inhibit key components of bacterial cell wall production that, when disrupted, consistently kill the cell.”
|
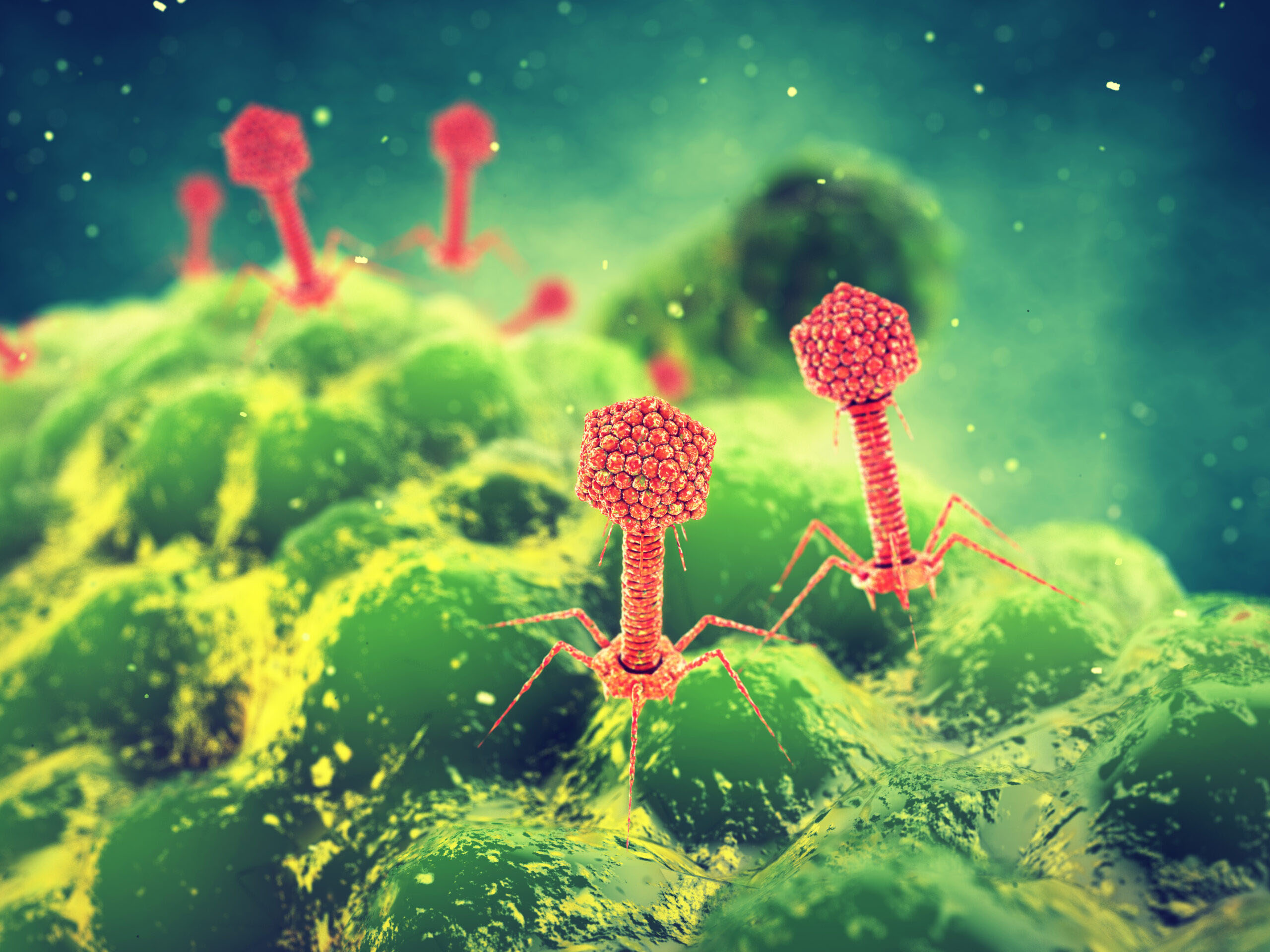 |
| A rendering of bacteriophage viruses attacking a bacteria. (Image: nobeastsofierce)
|
|
There appears to be at least one type of phage for every known strain of bacteria, and they are thought to be the most abundant biological entities on Earth. In fact, there are an estimated 1031 phage particles on the planet right now, or the equivalent of one trillion phages for every grain of sand. Each of these phages evolve alongside their chosen host strain, allowing them to counter bacterial resistance traits, as they arise, with improved biological weaponry.
|
|
This massive abundance, specificity, and efficacy means that there are plenty around to study, and that we should theoretically be able to use phages to control any harmful microbe. Phages are also harmless to non-bacterial cells, another reason they are so appealing as medicines and biocontrol tools.
|
|
The problem arises when trying to isolate a single phage from the environment and determine which microbe it targets and how. Scientists are often unable to assess phage-bacteria battles based on genomic sequence alone or study them in action because many bacteria can’t be cultured in a lab – and even if they could, there’s an inherent catch-22 of needing to know ahead of time which bacteria to culture in order to study the phages that infect and kill them.
|
|
To sidestep these obstacles and identify the cellular targets of Sgls, Mutalik and his colleagues used a technology the team previously invented called Dual-Barcoded Shotgun Expression Library Sequencing (Dub-seq). Dub-seq allows scientists to employ a coded library of DNA fragments to investigate how unknown genes function, and can be applied to complicated environmental samples that contain the DNA of many organisms – no culturing needed. In this study, the authors used six Sgls from six phages that infect different bacteria and identified the part of the bacterial cell wall or supporting molecules that each Sgl attacks. In collaboration with scientists from Texas A&M University, they conducted a detailed characterization of the function of one Sgl.
|
|
This work showed that the Sgl proteins target pathways for cell wall building that arose very early in the evolutionary history of bacteria and are still used by nearly all bacteria (including pathogenic bacteria). Since the Sgl proteins attack such fundamental and ubiquitous targets, they can kill bacteria other than the phage’s target strain – confirming they have great potential as antibiotics.
|
|
“Phages are extraordinary innovators when it comes to destroying bacteria. We’re really excited to uncover novel bacterial pathogen-targeting mechanisms that could be leveraged into therapies,” said first author Benjamin Adler, a postdoctoral fellow in Jennifer Doudna’s lab at UC Berkeley.
|
|
Now that the team has evaluated the Dub-seq approach for tackling this question, they can apply it to the thousands of single-gene lysis producing phages awaiting characterization in environmental samples that the team has collected from the ocean, soils, and even the human gut. The inspiration for the next breakthrough medicine could be in there, waiting.
|

