| Aug 10, 2023 |
Researchers use enzymes to develop a new class of materials for biocatalytic processes
|
|
(Nanowerk News) Industrial biocatalysis is mainly used for the production of pharmaceuticals and special chemicals. To advance this process, researchers are working on new process technologies. In biocatalysis, enzymes accelerate the reactions instead of chemical catalysts. This saves resources and energy.
|
|
Current work is aimed at continuously providing large quantities of enzyme biocatalysts under conditions that are as gentle as possible. To achieve efficient substance conversions, enzymes are immobilized in microstructured flow reactors. They are spatially fixed and bound to an inert material. Hence, their mobility is limited, which results in a higher concentration of enzymes and a higher productivity.
|
Foamed Microdroplets from Self-assembling Enzymes
|
|
Normally, foaming modifies the enzyme structure and enzymes lose their biocatalytic activity. The new protein foams, however, have a tremendous stability and activity. Activity is a measure of the effectiveness of the enzyme that ensures that the initial substances react with each other as quickly as possible. For the production of protein foams, two dehydrogenase enzymes are mixed, which carry matching sites to spontaneously form a stable protein network.
|
|
“Then, a gas flow is added to this mix in a microfluidic chip for the controlled formation of microscopic bubbles of uniform size,” explains Professor Christof Niemeyer from the Institute for Biological Interfaces 1. The foam with the uniform bubble size is applied directly onto plastic chips and dried, as a result of which the proteins polymerize and form a stable hexagonal lattice.
|
|
“These monodisperse all-enzyme foams are three-dimensional porous networks consisting of biocatalytically active proteins exclusively,” Niemeyer says. The stable hexagonal honeycomb structure of the foams has a mean pore diameter of 160 µm and a thickness of the lamellae of 8 µm. It is formed by the uniform spherical bubbles after a few minutes.
|
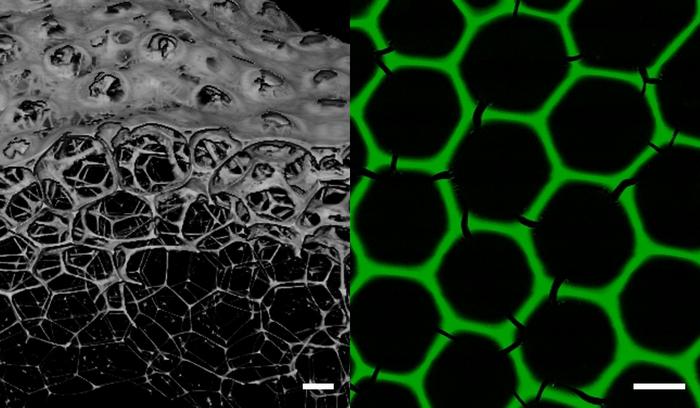 |
| New biomaterials for industrial biocatalysis: Enzyme foams form three-dimensional porous networks with a stable hexagonal honeycomb structure. (Image: Julian Hertel, KIT)
|
Efficient Use of the Active and Stable Full-enzyme Foams
|
|
To efficiently use the enzymes for conversion reactions, they have to be immobilized in large quantities under most gentle conditions in order to maintain their activity. So far, enzymes have been immobilized on polymers or carrier particles. However, these require valuable reactor space and enzyme activity may be reduced.
|
|
“Compared to our ‘all-enzyme hydrogels,’ the new foam-based materials have a far larger surface area on which the desired reaction can take place,” Niemeyer points out.
|
|
In contrast to theoretically expected results, the new foams surprisingly show strikingly high durability, mechanical resistance, and catalytic activity of the enzymes, which had not previously been achieved in foaming proteins. The researchers assume that stability results from the matching junctions of the enzymes. They enable the enzymes to self-assemble and to form a new material network of unmatched stability during drying.
|
|
“Surprisingly, the newly developed enzyme foams are far more stable after drying for four weeks than the same enzymes without foams,” Niemeyer says. “This is particularly useful for the commercialization of the process, as production on stock and delivery are facilitated considerably.”
|
|
The work has been published in (Advanced Materials, "Biocatalytic Foams from Microdroplet-Formulated Self-Assembling Enzymes").
|
|
The new biomaterials open up many opportunities for innovations in industrial bioengineering, materials sciences, or food technology. Protein foams might be used in biotechnological processes to produce valuable compounds more efficiently and sustainably. The researchers, for instance, used the foams to produce tagatose which is a promising alternative to refined sugar.
|

