| May 18, 2023 |
'Electrifying' moment for more sustainable polymers from low cost materials
|
|
(Nanowerk News) Organic chemistry researchers at Flinders University have ‘struck gold’ by discovering a new way to make ‘green’ polymers from low-cost building blocks with just a small amount of electricity.
|
|
The reaction is fast and occurs at room temperature. No hazardous chemical initiators are required – just electricity, with many potential uses including in gold mining and recycling e-waste, an interdisciplinary team reveal in an article just published in the Journal of the American Chemical Society ("Electrochemical synthesis of poly(trisulfides)").
|
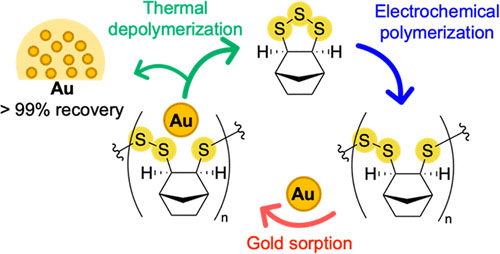 |
| Electrochemically initiated ring-opening polymerization of norbornene-based cyclic trisulfide monomers deliver well-defined, linear poly(trisulfides), which were solution processable. (© JACS)
|
|
“The use of electricity to produce new materials is an emerging field of research that opens many doors to new chemicals and polymers that can be produced in a more sustainable way,” says co-author Dr Thomas Nicholls, an expert in using electrochemistry to make valuable molecules.
|
|
The process begins by adding an electron to the basic building block or monomer. After ‘electrocuting’ the monomer, it reacts with another building block in a chain reaction which leads to the formation of a polymer.
|
|
First-author and PhD candidate Jasmine Pople says: “Our method to electrochemically produce polymers provides new materials that are highly functional and environmentally friendly.”
|
|
“The use of electricity to make valuable molecules is expanding rapidly due to its versatility. Additionally, it may generate less waste than traditional chemical syntheses and it can be powered with renewable energy.”
|
|
The key polymer made by the team has sulfur-sulfur bonds in its backbone. These sulfur groups can do useful things like bind precious metals such as gold. The team demonstrated that the key polymer could remove 97% of gold from solutions of relevance to mining and e-waste recycling.
|
|
The sulfur-sulfur bonds can also be broken and reformed. This interesting property enabled the team to discover conditions to convert the polymer back to its original building block. This is an important advance in recycling.
|
|
Typically, when common plastics are recycled, they are simply heated and reshaped into a new product. This process can cause degradation and down-cycling (conversion to a less valuable material), leading to eventual disposal in landfill.
|
|
In contrast, the polymers made in the latest research from Flinders University scientists can be chemically converted back into its constituent building blocks in high yield – meaning that building block can be used again to make new polymers.
|
|
The team also carried out quantum mechanical calculations to understand the details of how the reaction works. The findings were surprising and fortuitous.
|
|
“The polymerisation has a clever self-correcting mechanism: whenever the wrong reaction occurs, it reverses until the correct reaction proceeds, ensuring a uniform polymer,” says Research Associate in computational and physical chemistry Dr Le Nhan Pham.
|
|
Future applications of this class of materials include environmental remediation, gold mining, and use of the polymer as an anti-microbial agent.
|

