| Mar 28, 2013 |
Even graphene has weak spots
|
|
(Nanowerk News) Graphene, the single-atom-thick form of carbon, has become famous for its extraordinary strength. But less-than-perfect sheets of the material show unexpected weakness, according to researchers at Rice University in Houston and Tsinghua University in Beijing.
|
|
The kryptonite to this Superman of materials is in the form of a seven-atom ring that inevitably occurs at the junctions of grain boundaries in graphene, where the regular array of hexagonal units is interrupted. At these points, under tension, polycrystalline graphene has about half the strength of pristine samples of the material.
|
|
Calculations by the Rice team of theoretical physicist Boris Yakobson and his colleagues in China were reported this month in the American Chemical Society journal Nano Letters ("Pseudo Hall–Petch Strength Reduction in Polycrystalline Graphene"). They could be important to materials scientists using graphene in applications where its intrinsic strength is a key feature, like composite materials and stretchable or flexible electronics.
|
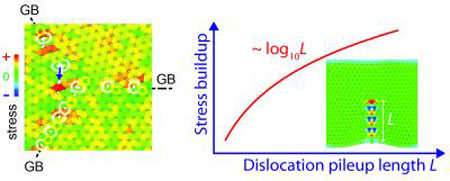 |
| New work by theorists at Rice and Tsinghua universities shows defects in polycrystalline forms of graphene will sap its strength. The illustration from a simulation at left shows a junction of grain boundaries where three domains of graphene meet with a strained bond in the center. At right is the calculated stress buildup at the tip of a finite-length grain boundary. (Image: Vasilii Artyukhov/Rice University)
|
|
Graphene sheets grown in a lab, often via chemical vapor deposition, are almost never perfect arrays of hexagons, Yakobson said. Domains of graphene that start to grow on a substrate are not necessarily lined up with each other, and when these islands merge, they look like quilts, with patterns going in every direction.
|
|
The lines in polycrystalline sheets are called grain boundaries, and the atoms at these boundaries are occasionally forced to change the way they bond by the unbreakable rules of topology. Most common of the "defects" in graphene formation studied by Yakobson's group are adjacent five- and seven-atom rings that are a little weaker than the hexagons around them.
|
|
The team calculated that the particular seven-atom rings found at junctions of three islands are the weakest points, where cracks are most likely to form. These are the end points of grain boundaries between the islands and are ongoing trouble spots, the researchers found.
|
|
"In the past, people studying what happens at the grain boundary looked at it as an infinite line," Yakobson said. "It's simpler that way, computationally and conceptually, because they could just look at a single segment and have it represent the whole."
|
|
But in the real world, he said, "these lines form a network. Graphene is usually a quilt made from many pieces. I thought we should test the junctions."
|
|
They determined through molecular dynamics simulation and "good old mathematical analysis" that in a graphene quilt, the grain boundaries act like levers that amplify the tension (through a dislocation pileup) and concentrate it at the defect either where the three domains meet or where a grain boundary between two domains ends. "The details are complicated but, basically, the longer the lever, the greater the amplification on the weakest point," Yakobson said. "The force is concentrated there, and that's where it starts breaking."
|
|
"Force on these junctions starts the cracks, and they propagate like cracks in a windshield," said Vasilii Artyukhov, a postdoctoral researcher at Rice and co-author of the paper. "In metals, cracks stop eventually because they become blunt as they propagate. But in brittle materials, that doesn't happen. And graphene is a brittle material, so a crack might go a really long way."
|
|
Yakobson said that conceptually, the calculations show what metallurgists recognize as the Hall-Petch Effect, a measure of the strength of crystalline materials with similar grain boundaries. "It's one of the pillars of large-scale material mechanics," he said. "For graphene, we call this a pseudo Hall-Petch, because the effect is very similar even though the mechanism is very different.
|
|
"Any defect, of course, does something to the material," Yakobson said. "But this finding is important because you cannot avoid the effect in polycrystalline graphene. It's also ironic, because polycrystals are often considered when larger domains are needed. We show that as it gets larger, it gets weaker.
|
|
"If you need a patch of graphene for mechanical performance, you'd better go for perfect monocrystals or graphene with rather small domains that reduce the stress concentration."
|

