| Sep 24, 2013 |
Non-precious metal catalysts outperforming platinum-based ones
|
|
(Nanowerk News) Researchers from Ulsan National Institute of Science and Technology (UNIST), Korea Institute of Energy Research (KIER), and Brookhaven National Laboratory, have discovered a new family of non-precious metal catalysts. These catalysts exhibit better performance than platinum in oxygen-reduction reaction (ORR) only with 10% of the production cost of a platinum catalyst. The finding, described in Nature's Scientific Reports ("Ordered mesoporous porphyrinic carbons with very high electrocatalytic activity for the oxygen reduction reaction"), provides an important step towards circumventing the biggest obstacle to widespread- commercialization of fuel cell technology.
|
|
Fuel cells have various advantages compared to internal combustion engines or batteries, due to their high energy conversion efficiency and environmentally benign and quiet operation conditions. However, the high cost and instability of platinum catalysts for oxygen reduction reaction at the cathode have critically impeded the extensive application of polymer electrolyte fuel cells.
|
|
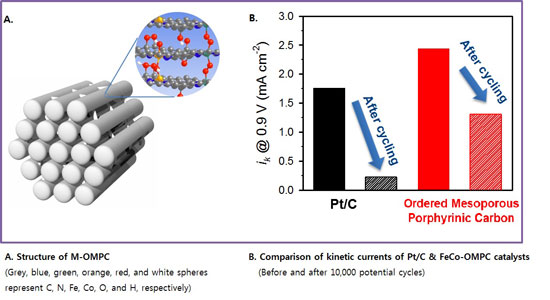
The UNIST research team reported on a new family of non-precious metal catalysts based on ordered mesoporous porphyrinic – any of a class of heterocyclic compounds containing four pyrrole rings arranged in a square – carbons (M-OMPC) with high surface areas and tunable pore structures.
|
 |
|
"Our synthetic strategy for the non-precious metal catalysts included a multitude of advantages that would be favorable to PEFC applications" said Prof. Joo. "First, our synthetic method is amenable to simple and mild experimental conditions. Second, the synthesis of the M-OMPC catalysts could be readily scaled up to a few tens of grams in a single batch. Third, well-developed, hierarchical micro-mesoporosity would be advantageous for efficient transport of fuels and by-products. Finally, the M-OMPC catalysts showed very high surface areas, which could significantly increase the density of the catalytically active sites accessible to reactants."
|
|
The research was led by Sang Hoon Joo, professor of the School of Nano-Bioscience and Chemical Engineering at South Korea's UNIST. Fellow authors include: Jae Yeong Cheon from UNIST; Gu-Gon Park from the Korea Institute of Energy Research (KIER); Radoslav R. Adzic from the Chemistry Department of the Brookheaven National Laboratory.
|
|
The materials developed by the UNIST research team were prepared by nanocasting ordered mesoporous silica (OMS) templates with metalloporphyrin precursors. In addition they were constructed with three dimensional networks of porphyrinic carbon frameworks.
|
|
The best M-OMPC catalyst showed an extremely high electrocatalytic activity for ORR in an acidic media. Its ORR activity is one of the best among the non-precious metal catalysts ever, and even higher than the state-of-the-art Pt catalyst. In addition, the FeCo-OMPC showed superior long-term durability and methanol-tolerance in ORR, compared to the Pt catalyst.
|
|
The research team attributed the high ORR activity of the FeCo-OMPC to its relatively weak interaction with oxygen as well as the high surface area design of the catalyst.
|
|
"Currently the world is striving to look for another energy source for increased energy demand and environmental issue," said Prof. Joo. "The novel material developed by the UNIST research team would be a solution to commercialize the eco-friendly and cost-effective fuel cells."
|

