| Jan 22, 2014 |
From a carpet of nanorods to a thin film solar cell absorber within a few seconds
|
|
(Nanowerk News) Research teams at the Helmholtz-Zentrum Berlin für Materialien und Energie (HZB) and at the University of Limerick, Ireland, have discovered a novel solid state reaction which lets kesterite grains grow within a few seconds and at relatively low temperatures. For this reaction they exploit a transition from a metastable wurtzite compound in the form of nanorods to the more stable kesterite compound. At the EDDI Beamline at BESSY II, the scientists could observe this process in real-time when heating the sample: in a few seconds Kesterite grains formed. The size of the grains was found to depend on the heating rate. With fast heating they succeeded in producing a Kesterite thin film with near micrometer-sized crystal grains, which could be used in thin film solar cells. These findings have now been published in the journal Nature Communications ("Phase-transition-driven growth of compound semiconductor crystals from ordered metastable nanorods").
|
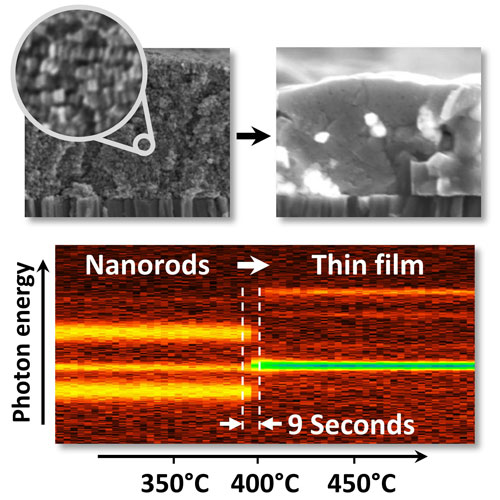 |
| The transformation from a layer of closely packed nanorods (top left) to a polycrystalline semiconductor thin film (top right) can be observed in by in-situ X-ray diffraction in real time. The intensities of the diffraction signals are color coded in the image at the bottom. A detailed analysis of the signals reveals that the transformation of the nanorods into kesterite crystals takes only 9 to 18 seconds. (Image: R. Mainz/A. Singh)
|
|
Grain formation during growth of kesterite solar cells observed in real-time
|
|
As starting material for the formation of the kesterite film serves a “carpet of nanorods”: With the help of solution-based chemical processing, the chemists around Ajay Singh and Kevin Ryan at the University of Limerick have fabricated films of highly ordered wurtzite nanorods, which have exactly the same composition as kesterite Cu2ZnSnS4. With the help of real-time X-ray diffraction at the EDDI beamline of BESSY II, HZB physicists around Roland Mainz and Thomas Unold could now observe how a phase transition from the metastable wurtzite phase to the stable kesterite phase leads to a rapid formation of a thin film with large kesterite grains.
|
|
“It is interesting to see that the complete formation of the kesterite film is so fast”, says Mainz. And the faster the samples are heated up, the larger the grains grow. Mainz explains that at low heating rate, the transition from wurtzite to kesterite starts at lower temperature at which many small grains form – instead of a few larger grains. Additionally, more defects are formed at lower temperatures. During fast heating, the transition takes place at higher temperature at which grains with less defects form.
|
|
Moreover, the comparison of the time-resolved evolution of the phase transition during slow and during fast heating shows that not only the grain growth is triggered by the phase transition, but also the phase transition is additionally accelerated by the grain growth. The HZB physicists have developed a model which can explain these findings. By means of numerical model calculations, they demonstrated the accordance of the model with the measured data.
|
|
Novel synthesis pathway for thin film semiconductors with controlled morphology
|
|
The work points towards a new pathway for the fabrication of thin microcrystalline semiconductor films without the need of expensive vacuum technology. Cu2ZnSnS4-based kesterite semiconductors have gained increasing attention in the past, since they are a promising alternative for the Cu(In,Ga)Se2 chalcopyrite solar cells which already achieved efficiencies above 20%. Kesterite has similar physical properties as the chalcopyrite semiconductors, but consist only of elements which are abundantly present in the earth crust.
|
|
The new procedure could also be interesting for the fabrication of micro- and nanostructured photoelectric devices as well as for semiconductor layers consisting of other materials, says Mainz. “But we continue to focus on kesterites, because this is a really exciting topic at the moment.”
|

