| Posted: Dec 08, 2010 | |||||
Can nanoparticles end up in the brain? |
|||||
| (Nanowerk Spotlight) The use of minute particles as drug carriers for targeted therapy has been studied and discussed for more than 20 years. A selective accumulation of active substances in target tissues has been demonstrated for certain so-called nanocarrier systems that are administered bound to pharmaceutical drugs. Great expectations are placed on nanocarrier systems that can overcome natural barriers such as the blood-brain barrier (BBB) and transport the medication directly to the desired tissue and thus heal neurological diseases that were formerly incurable. The BBB represents the border between the circulating blood and the fluid in the central nervous system. It functions to protect the sensitive nerve cells from foreign substances and infections from the blood. Whether nanoparticles enter the central nervous system unintentionally and induce health problems is also being debated. | |||||
| This dossier illustrates how the BBB functions and the positive effects of the new therapy possibilities, but also discusses the negative impacts of nanoparticles that enter the brain unintentionally. | |||||
| The blood-brain barrier | |||||
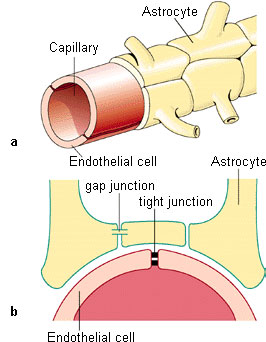
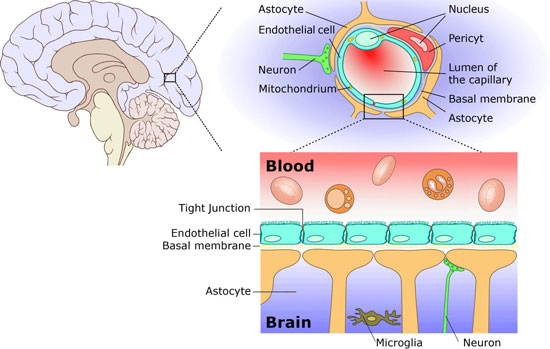
| The brain is permeated with a network of fine blood vessels. These so-called capillaries supply the brain with nutrients and oxygen. Combined, the walls of these blood vessels form the so-called blood-brain barrier. In all mammals, as well as in humans, it serves as a physiological barrier between the blood circulation system and the brain. Its task is to protect the brain from diseasecausing agents, toxins and messenger substances circulating in the blood. The BBB therefore represents a highly selective filter through which the nutrients needed by the brain pass in one direction and the resulting metabolic wastes in the other. This supply and removal involves a series of special transport processes. Within the central nervous system, the spaces between the neurons (nerve cells) are almost entirely filled by glia or endothelial cells and their processes (Figure 1). | |||||
 |
|||||
| Figure 1: The blood-brain barrier; above, cross section through the brain; center, schematic representation of the BBB; below, cellular structure. (Source: Ref. 11) | |||||
The metabolism of the nerve cells is via these
endothelial cells. These cells serve to insert,
isolate and supply the nerve cells and their
neurons. One type of glia cells are the astrocytes
(Figure 2). They bear numerous processes
with which they attach themselves to
the walls of the capillaries and form a nearly
uninterrupted coating surrounding the
capillaries. One feature distinguishes the vessel
walls of the capillaries that form the
blood-brain barrier from other blood vessels
in the body: there is a solid connection
between the adjoining vessel wall cells (socalled
capillary endothelial cells).
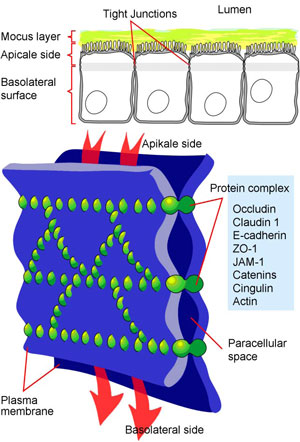
Tight junctions therefore have three key functions: i) a barrier function by closing up the intercellular spaces, ii) the mechanical stabilization of the epithelial cell aggregates and iii) maintaining the polarity of the epithelial cells by preventing the free floating of membrane components along the cell membranes. Substances that need to enter the brain from the blood or move from the brain into the blood cannot circumvent these cells, but must be channeled directly through the vessel wall cells by special transport systems (Figure 4). This controlled process enables a selective exchange of substances between nerve cells and blood, and protects the nerve cells from penetration by harmful substances. Those substances that are necessary for supplying the brain can pass unhindered, namely oxygen and carbon dioxide. At the same time, specific transport systems transfer D-glucose, D-hexose, several L-amino acids and certain lipid-soluble substances through the BBB. This is accompanied by a release of degradation products into the blood. The terminal processes of the astrocytes represent a barrier for numerous substances such as certain hormones, non-lipid-soluble, water-soluble and chemical substances, along with proteins, which helps maintain a constant milieu for the neurons of the nervous system.
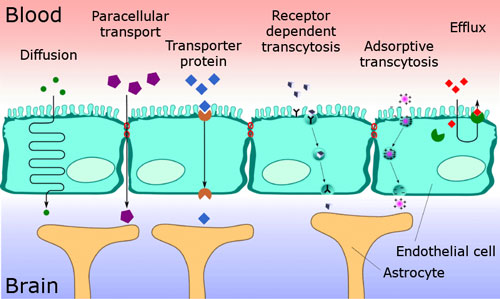
As noted above, the BBB ? despite its function as a protective barrier ? must also enable the transport of nutrients to the brain and the corresponding removal of metabolic products. Accordingly, water-soluble substances and peptides overcome this barrier via specific transporters or special canals in the cell membrane (diffusion, paracellular transport, specific transporter proteins, receptor- mediated transport, adsorptive transport, see Figure 4), while the other soluble compounds pass this barrier via passive diffusion. Nanoparticles and the blood-brain barrier: An opportunity for the treatment of diseases – are there associated risks? |
|||||
| Nanoparticles can be used as carrier systems to overcome the BBB and deliver specific medications to regions of the brain that would normally be inaccessible. Their surfaces can be coated with certain materials or manufactured such that they can pass the BBB and transport the pharmaceuticals to the sites where they are needed. Thus, nanoparticle carrier systems can distribute medications spatially and temporally in the brain and help cure diseases that were previously untreatable. This enables a tissue-specific accumulation of drugs, special depot effects and overcomes the body's own barriers such as the blood-brain barrier. This would allow stronger concentrations of medications to be applied with improved effects. This is a booming research and development field and shows ever greater promise in treating diseases such as Alzheimer's, Parkinson's or certain brain tumors. Currently, most applications of nanoparticle carrier systems involve drugs that are already in clinical use. Research is being conducted on various scenarios for the application of nanoparticle carrier systems. For example, experiments are being performed on rats to determine whether the direct transport of genes (gene therapy) is possible into specific regions of the brain via nanocarriers in order to replace damaged hereditary substances and therefore treat Parkinson's disease, for example1. | |||||
 |
|||||
| Figure 4: Various transport pathways through the BBB. (Source: Ref. 14) | |||||
| One currently used form of therapy, although it does not overcome the BBB, is the so-called hyperthermia therapy, which uses nano iron particles to treat brain tumors such as glioblastoma. Here, magnetized, iron-containing nanoparticles are injected directly into the brain tumor. These nanoparticles, also known as "Trojan horses", are actively taken up by the tumor cells because they are coated with a layer of sugar molecules2. This approach packs the cancer cells full of ironcontaining particles, which can then be pinpointed and heated by electromagnetic fields. This overheating kills the tumor cells. What remains unclear is the fate of the nanoparticles remaining in the brain, what effect they have there or whether they are eliminated3; 4. | |||||
| An in-vitro study5 showed that TiO2-nanoparticles can trigger oxidative stress6, especially in certain brain cells, namely in the socalled microglia cells (phagocyting immune cells of the central nervous system). This effect is only possible if the BBB is crossed. In vivo studies have been unable to definitively confirm this. In experiments on rats, injecting TiO2-nanoparticles directly into the blood stream did not lead to accumulation of the particles in the brain7. A more recent study, however, shows that relatively high concentrations of TiO2-nanoparticles injected into pregnant mice were detectable in the brain of the offspring8. A further study by the same research group revealed changes in the DNA (gene expression) in the brain tissue of the fetuses9. In this case, the BBB as well as the barrier between the mother and placenta may have been breached. Another mechanisms beyond crossing physiological barriers might be at play here because, during the embryonic phase, the BBB is not yet fully developed. The researchers point out, however, that the determined effects are not directly transferable to humans. In particular, the biological relevance of the described gene alterations is unclear. Unfortunately, there are still too few results to draw definitive conclusions on the permeability of TiO2-nanoparticles through the blood-brain barrier. | |||||
| Another route by which nanoparticles can enter the brain is via the olfactory nerve, circumventing the blood-brain barrier. The olfactory nerve (nervus olfactorius) has a direct connection to the brain by way of its long processes (axons). It is therefore conceivable that inhaled nanoparticles enter the olfactory nerve and are transported to the brain along the axons. This phenomenon has been observed in rats. After inhalation, individual carbon-containing nanoparticles entered the olfactory bulbs in the nose and were then transported along the olfactory nerve to the brain10. The extent to which this result is transferable to humans remains unclear because the brain anatomy of rats and humans differs considerably. In this case as well, no final conclusions can be drawn as to whether the concentrations of nanoparticles transported in this manner – which are probably minimal – is biologically relevant and therefore relevant from a health perspective. Health effects would be expected only under continuous exposure and exposure to high concentrations of nanoparticles. | |||||
| Conclusions | |||||
| Artificially manufactured particles can be applied to help overcome natural physiological barriers such as the blood-brain barrier. This phenomenon can be used to intentionally transport drugs to parts of the organism where they are needed, for example the brain. Research is currently being conducted to determine whether nanoparticles are able to reach the brain by other mechanisms such as along the olfactory nerve. It also remains poorly known whether nanoparticles unintentionally pass the blood-brain barrier and cause potential damage. The few available studies on the risks of nanoparticles that have entered the central nervous system are controversial. This prevents drawing definitive conclusions about the health effects of unintentional exposure of the brain to nanoparticles. | |||||
| Notes and References | |||||
| 1 Zhang, Y., Calon, F., Zhu, C., Boado, R. J. and Pardridge, W. M., 2003, Intravenous nonviral gene therapy causes normalization of striatal tyrosine hydroxylase and reversal of motor impairment in experimental parkinsonism, Hum Gene Ther 14(1), 1-12 | |||||
| 2 Simk?, M., Fiedeler, U., Gazs?, A. and Nentwich, M., 2008, Einfluss von Nanopartikeln auf zellul?re Funktionen. NanoTrust-Dossiers 007 epub.oeaw.ac.at/ita/nanotrust-dossiers/dossier007.pdf. | |||||
| 3 Jordan, A. and Maier-Hauff, K., 2007, Magnetic nanoparticles for intracranial thermotherapy, J Nanosci Nanotechnol 7(12), 4604-6 | |||||
| 4 Jordan, A., Scholz, R., Maier-Hauff, K., van Landeghem, F. K., Waldoefner, N., Teichgraeber, U., Pinkernelle, J., Bruhn, H., Neumann, F., Thiesen, B., von Deimling, A. and Felix, R., 2006, The effect of thermotherapy using magnetic nanoparticles on rat malignant glioma, J Neurooncol 78(1), 7-14 | |||||
| 5 Long, T. C., Saleh, N., Tilton, R. D., Lowry, G. V. and Veronesi, B., 2006, Titanium dioxide (P25) produces reactive oxygen species in immortalized brain microglia (BV2): implications for nanoparticle neurotoxicity, Environ Sci Technol 40(14), 4346-52 | |||||
| 6 Simk?, M., Gazs?, A., Fiedeler, U. and Nentwich, M., 2009, Nanopartikel, Freie Radikale und Oxidativer Stress, NanoTrust-Dossiers 012 epub.oeaw.ac.at/ita/nanotrust-dossiers/dossier012.pdf. | |||||
| 7 Fabian, E., Landsiedel, R., Ma-Hock, L., Wiench, K., Wohlleben, W. and van Ravenzwaay, B., 2008, Tissue distribution and toxicity of intravenously administered titanium dioxide nanoparticles in rats, Arch Toxicol 82(3), 151-7 | |||||
| 8 Takeda, K., Suzuki, K., Ishihara, A., Kubo-Irie, M., Fujimoto, R., Tabata, M., Oshio, S., Nihei, Y., Ihara, T. and Sugamata, M., 2009, Nanoparticles transferred from pregnant mice to their offspring can damage the genital and cranial nerve systems, J Health Sci 55, 95-102. | |||||
| 9 Shimizu, M., Tainaka, H., Oba, T., Mizuo, K., Umezawa, M. and Takeda, K., 2009, Maternal exposure to nanoparticulate titanium dioxide during the prenatal period alters gene expression related to brain development in the mouse, Part Fibre Toxicol 6, 20 | |||||
| 10 Oberdorster, G., Sharp, Z., Atudorei, V., Elder, A., Gelein, R., Kreyling, W. and Cox, C., 2004, Translocation of inhaled ultrafine particles to the brain, Inhal Toxicol 16(6-7), 437-45. | |||||
| 11 n.a. | |||||
| 12 edoc.hu-berlin.de/dissertationen/warmuth-carsten-2003-05-19/HTML/Warmuth_html_39865938.png. | |||||
| 13 n.a. | |||||
| 14 www.neuro24.de/show_glossar.php?id=288. | |||||
| By NanoTrust, Austrian Academy of Sciences. NanoTrust Dossiers are published irregularly and contain the research results of the Institute of Technology Assessment in the framework of its research project NanoTrust. The Dossiers are made available to the public exclusively on epub.oeaw.ac.at/ita/nanotrust-dossiers. | |||||
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|||||