| Posted: Jun 14, 2013 | |
Destroying cancer cells by remote activation of intracellular nanoparticles |
|
| (Nanowerk Spotlight) Under an applied magnetic field, iron oxide nanoparticles trigger cancer cell death by bursting intracellular organelles, according to research published in the May 21, 2013 online edition of ACS Nano ("Lysosomal Membrane Permeabilization by Targeted Magnetic Nanoparticles in Alternating Magnetic Fields"). | |
| “These findings give us a new strategy to treat cancer using nanomaterials,” explains Dr. Carlos Rinaldi, Professor of Chemical Engineering at the Universities of Florida and Puerto Rico and lead author on the study. “We may be able to administer magnetic nanoparticles externally, allow them to accumulate at the tumor site, and then irradiate them with a magnetic field to induce cancer cell death.” | |
| In a previous study ("EGFR-Targeted Magnetic Nanoparticle Heaters Kill Cancer Cells without a Perceptible Temperature Rise"), Rinaldi and his co-workers found that exciting iron oxide nanoparticles inside cancer cells with an alternating magnetic field caused the cells to undergo programmed cell death, known as ‘apoptosis’. However, the mechanism by which the iron oxide nanoparticles induced apoptosis was not clear. | |
| “Originally, we thought that applying the magnetic field would heat up the cancer cells and trigger cell death,” explains Rinaldi. “But, to our surprise, we found that cell death occurred without an overall increase in the temperature of the cells.” | |
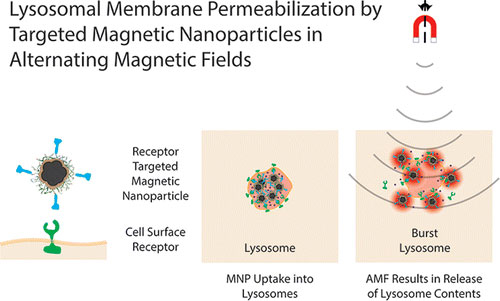
| When Rinaldi and his co-workers, including Maribella Domenech, Illeana Marrero-Berrios, and Madeline Torres-Lugo at the University of Puerto Rico, incubated iron oxide nanoparticles targeting the epidermal growth factor receptor with cancer cells, they found that the nanoparticles were internalized in small intracellular vesicles called ‘endosomes’. The endosomes later fused with ‘lysosomes’, a special type of organelle that contains digestive enzymes and an acidic environment. | |
| “Instead of killing cells by hyperthermia, we suspected that exciting the iron oxide nanoparticles caused the lysosomes to rupture, releasing lysosomal enzymes,” explains Rinaldi. If lysosomal enzymes are released into the cell cytoplasm, they can trigger cell death by apoptosis. | |
| Rinaldi and his team incubated breast cancer cells with the iron oxide nanoparticles and a fluorescent dye that changes color when it interacts with intact lysosomes. After subjecting the cells to an alternating magnetic field, the fraction of intact lysosomes decreased by a factor of 3. The team also found that a key lysosomal enzyme, ‘cathepsin B’, localized to the cell cytoplasm. At the same time, there was a significant increase in the intracellular production of reactive oxygen species (ROS), which is a hallmark of apoptotic cell death. Nanoparticles without a targeting ligand did not induce significant changes in the number of lysosomes, localization of cathepsin B, or production of ROS. | |
| While these results confirmed the initial hypothesis, Rinaldi points out that is not clear exactly how exciting the iron oxide nanoparticles with the magnetic field permeabilized the lysosomes. “The magnetic field not only causes the nanoparticles to heat their environment, it also causes them to rotate rapidly. Either, or both, of these factors could disrupt the lysosomal membrane.” More studies are needed to tease out the mechanism, which is important for controlling the effect. | |
 |
|
| Following excitation by an alternating magnetic field, iron oxide nanoparticles induce cell death by bursting intracellular lysosomes. (Reprinted with permission. Copyright 2013, American Chemical Society) | |
| In the past, antibodies and small molecules were used to trigger apoptosis in cancer cells. However, cancer cells often adapt to resist these treatments. Because iron oxide nanoparticles cause physical damage to cancer cells, it is difficult for them to develop resistance. | |
| The other problem with pro-apoptotic drugs is that they often kill healthy cells in addition to cancerous cells, leading to off-target toxicity. Rinaldi’s strategy avoids off-target toxicity in two ways. First, the nanoparticles are functionalized with the epidermal growth factor (EGF) peptide. The receptor for EGF is over-expressed on certain form of cancer. As a result, the nanoparticles are internalized preferentially by the cancer cells, and not by healthy cells. Second, the nanoparticles only trigger apoptosis when they are excited by an external magnetic field. By applying the magnetic field to affected areas of the body, toxicity in healthy tissues can be avoided. | |
| In addition to triggering cell death, permeabilization of the lysosomes may aid the delivery of drugs and other therapeutics to cancer cells. Many researchers have attempted to use nanoparticles as drug carriers. However, the payload is often trapped inside endosomes and/or lysosomes. To promote endosomal escape, researchers often modify nanoparticles with functional groups that destabilize lysosomal membranes either by creating an osmotic gradient or by directly interacting with the membrane. While these modifications can promote the delivery of a cargo within the cell cytoplasm, they often cause toxicity in healthy tissues. “Using magnetic nanoparticles as delivery vehicles, we can release cargo from the lysosomes by applying a magnetic field. This allows us to control therapeutic release in both space and time,” explains Rinaldi. | |
| Although this strategy is not ready for clinical applications in human patients, Rinaldi is confident in its prospects. “Iron oxide nanoparticles are already used clinically and we know that they are safe.” On top of this, the radio-frequency fields used to excite the nanoparticles can penetrate deep within tissues, so tissue attenuation should not be a problem. As a next step towards eventual clinical translation, Rinaldi and his co-workers plan on using the iron oxide nanoparticles to treat tumors in small animal models. | |
|
By Carl Walkey, Integrated Nanotechnology & Biomedical Sciences Laboratory, University of Toronto, Canada.
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
