| Posted: Oct 12, 2016 | |
A multifunctional biophotonic platform enabled by moire metasurfaces |
|
| (Nanowerk Spotlight) Biophotonics has recently captured strong interests from both academic and industry due to the high potential of applying photonic technologies to biomedical applications, including cell manipulation and biological analysis. | |
| In particular, plasmonic metasurfaces, benefiting from their extraordinary capabilities in confining and manipulating electromagnetic waves, have been successfully utilized in photothermal therapy and detecting biomolecules with unprecedented low concentration. | |
| Although a variety of designs have been reported for different applications, it remains challenging to achieve one metasurface that can be applied as an integrated biophotonic platform for multiple applications. | |
| This limitation is mainly due to the different operation wavelength regimes – visible, near-infrared, and mid-infrared, etc. – required for different applications. Nowadays, multifunctional biophotonic platforms that are simultaneously capable of several biomedical functions, such as spectroscopy analysis, in-situ therapy, and micro-manipulations are highly desired. | |
| Researchers at the University of Texas at Austin, led by professors Yuebing Zheng, Gennady Shvets, Deji Akinwande and Seth Bank, demonstrate for the first time a multifunctional biophotonic platform enabled by the multiband resonance peaks of the plasmonic moiré metasurfaces. | |
| The results of this work have been published in the October 7, 2016 online edition of Nanoscale ("Dual-Band moiré Metasurface Patches for Multifunctional Biomedical Applications"). | |
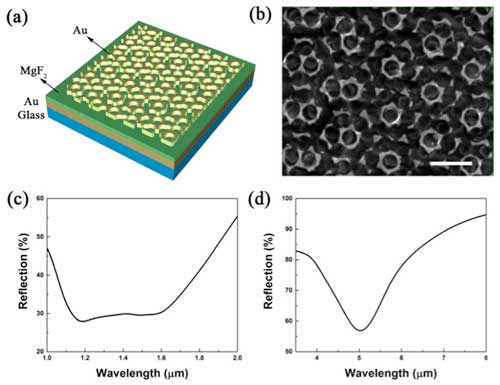
| "In our work, a sandwich-like metal-insulator-metal (MIM) structure is created, where a gold moiré metasurface is placed on top of a gold thin film with a MgF2 layer as a spacer between them," Zilong Wu, a researcher at University of Texas at Austin, and the paper's first author, tells Nanowerk. "Benefiting from the multiband nature of moiré metasurface and the near-field enhancement from the MIM configuration, a dual-band metasurface patch with strong plasmonic resonances at both near-infrared (∼1300 nm in wavelength) and mid-infrared (∼5 µm in wavelength) regimes was achieved." | |
| Wu explains that the metasurface fabricated by moiré nanosphere lithography, a cost-effective fabrication method to obtain moiré patterns, consists of plasmonic nanostructures that support plasmon resonances at different wavelengths due to the gradient in size and shape (read a previous Nanowerk Spotlight on nanosphere lithography as a novel technique to fabricate tunable plasmonic metasurfaces). | |
 |
|
| Figure 1. (a) Schematic of gold moiré metasurface patch (AMMP). (b) Scanning electron micrograph of a gold moiré metasurface of the AMMP. The scale bar is 2 µm. (c) Reflection spectrum of an AMMP in NIR regime. (d) Reflection spectrum of an AMMP in MIR regime. (© Royal Society of Chemistry) | |
| The broadband absorption in near-infrared (NIR) regime enables the high versatility in choosing working lasers with wavelengths from 1.2 to 1.6 µm for the photothermal treatment. | |
| In addition, the gold moiré metasurface patch is also promising for mid-infrared (MIR) sensing due to the strong plasmonic resonance at ∼5 µm. | |
| The researchers have made use of the dual-band properties of this plasmonic platform for several biomedical applications, including infrared sensing, optical capture and immobilization of bacteria, photothermal denaturation and spectroscopic analysis of proteins. | |
| Maruthi Yogeesh, a graduate student in UT Austin, and a co-author of this paper, notes that the strong absorption in NIR regime is highly promising for plasmon-assisted laser heating. | |
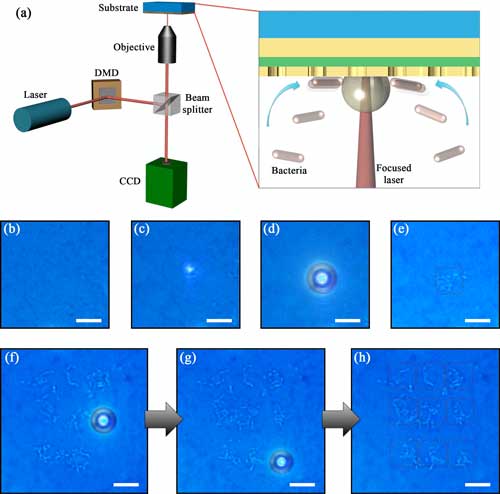
| "We immersed the metasurface patch in a solution containing bacteria and then shined a NIR laser on the substrate," he explains. "The plasmon-assisted heating effect quickly increases the localized temperature at the laser spot and generates a micro-bubble that can capture the suspending bacteria to the substrate." | |
 |
|
| Figure 2. (a) Schematic of the experimental setup for capture and immobilization of bacteria on the AMMP using plasmon-induced microbubbles. The inset shows the cross-sectional view of the transport and trapping of the bacteria at a microbubble generated by a laser beam at the interface of the AMMP and solution. (b) Optical images of AMMP substrate immersed in E.coli solution. (c) Optical image of AMMP irradiated by a NIR laser beam. (d) Optical image of a microbubble generated at the laser spot on the AMMP surface. (e) Optical image of E.coli cells immobilized on the AMMP after the disappearance of the bubble. (f-h) Time-evolved optical images show the microbubble-based patterning of 3 X 3 array of E.coli clusters (indicated by dashed squares) on the AMMP substrate. The patterning is from top to bottom and from left to right. The scale bars are 10 µm. (© Royal Society of Chemistry) (click on image to enlarge) | |
| The optical capturing of bacterial cells can be an effective approach for water sterilization. Compared with other sterilization methods based on photothermal effects, which require pre-deposition of bacteria on plasmonic substrates or plasmonic nanoparticles suspended in water, this microbubble-based method is simpler and 'greener'. | |
| The photothermal effect at NIR regime can be further combined with the strong plasmonic resonance at MIR regime to denaturalize proteins and subsequently perform in situ spectroscopy analysis. | |
| "Denaturation of protein through plasmonic heating effect has proven effective for cell inactivation applications," concludes Prof. Zheng. "Conventional characterization methods such as FTIR and fluorescence spectroscopies for the denaturation process require a large amount of samples or fluorescent labeling chemistry of proteins. Our gold moiré metasurface patch that provides photothermal treatment with in situ molecular characterization is promising as an integrated and cost-effective biomedical platform for both scientific research and clinical applications." | |
|
Provided by The University of Texas at Austin
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
