| Posted: Jan 12, 2017 | |
Topological defects make metal-free nanocarbon promising for zinc-air batteries |
|
| (Nanowerk Spotlight) The oxygen reduction reaction (ORR) is the core process – but also the bottleneck – for the cathode reaction of energy-conversion devices like certain types of fuel cells and metal-air batteries. | |
| "In spite of substantial advances, ORR electrocatalysis greatly suffers from its complex pathway, high overpotential, sluggish kinetics, and poor durability, thereby limiting the energy efficiency and power capability of prototype devices," says Prof. Qiang Zhang, a researcher from Tsinghua University, China, leading a group focused on advanced materials for efficient electrocatalysis and energy storage. "Nanocarbon materials are very promising alternatives for the noble metal catalysts, especially platinum, that have been used to boost this reaction. But previous works haven't yet figured out the exact active sites in nanocarbon materials and their specific roles during ORR and oxygen evolution reaction (OER). It makes our research trial and error." | |
| Recently, Prof. Zhang and co-workers from Tsinghua University, Queen Mary University of London, and Institute of Metal Research, Chinese Academy of Sciences, reported a novel heteroatom-doped and edge-rich graphene mesh and revealed the critical importance of edge and topological defects in the activity origin of metal-free nanocarbon materials for oxygen electrocatalysis, both experimentally and theoretically (Advanced Materials, "Topological Defects in Metal-Free Nanocarbon for Oxygen Electrocatalysis"). | |
 |
|
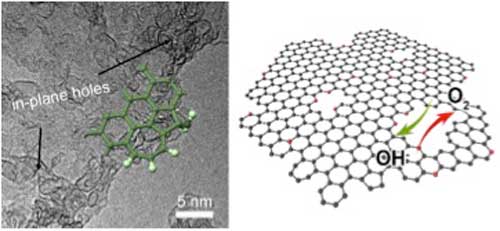
| Nitrogen-doped graphene mesh for ORR/OER. (Image: Tsinghua University) | |
| "We developed a very cheap and scalable process to fabricate graphene with nitrogen doping and topological defects," Cheng Tang, the first author explains. "Direct carbonization of a ternary composite with sticky rice, melamine, and Mg(OH)2 templates leads to few-layered graphene with abundant in-plane holes." | |
| Impressively, a high current density approaching 6.0 mA cm-2 and a low overpotential similar to commercial 20 wt% Pt/C can be obtained in both alkaline and acidic conditions using this graphene catalyst. In addition, the durability is superior without any obvious current change after 10 h test even in 1.0 M KOH. | |
| A primary Zn-air battery constructed using this catalyst can deliver a current density of ~6.0 mA cm-2 and a peak power density of ~3.0 mW cm-2. Besides, a remarkable OER activity comparable with Ir/C is obtained with a low overpotential gap of 0.90 V between ORR and OER, making it the best bifunctional metal-free catalysts ever reported. | |
| "With more control tests, we found that the pore-derived topological defects seem to be more important than the nitrogen doping for a higher activity, different from previous reports," says C. Tang. "Then we try to support our idea using density function theory calculations." | |
| Calculation results show that the N-doped moieties present a higher overpotential than pristine graphene nanoribbons, but those moieties with five-carbon ring or seven-carbon ring defects exhibit more enhanced activities. A nitrogen-free configuration with adjacent pentagon and heptagon carbon rings is revealed to exhibit the lowest overpotential for both ORR and OER. | |
| "It is different from previous results, but important for a thorough understanding of the activity origins of nanocarbon catalysts," notes Prof. Zhang. | |
| After a comprehensive review of recent advances probing the activity origins and underlying mechanism, including dopants, edges, and defects in carbocatalysis, Prof. Zhang and co-workers found that despite different active sites, they can modify the charge/spin distribution on the sp2-conjugated carbon matrix analogously, leading to optimized intermediate chemisorption and facilitated electron transfer. | |
| This work has been published in the January 9, 2017 online edition of Advanced Materials ("Nanocarbon for Oxygen Reduction Electrocatalysis: Dopants, Edges, and Defects"). | |
| As next steps, the scientists will move to further elucidate this point and improve the performances of such graphene catalyst towards practical applications in metal-air batteries or fuel cells. | |
| "Specific heteroatom doping at defective edges is expected to be the most effective strategy to alter electron structures and achieve optimal ORR activities for metal-free electrocatalysts," concludes Prof. Zhang. | |
|
Provided by Tsinghua University as a Nanowerk exclusive
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
