| Posted: Jan 30, 2017 | |
The formation of stable solid electrolyte interphases on lithium metal anode |
|
| (Nanowerk Spotlight) The successful commercialization of Li-ion batteries has promoted the fast development of electronics industry since 1990s. However, the current energy density cannot cater the demand of electric vehicles, wearable devices and smart grid. | |
| The energy density of Li-ion batteries based on intercalation-type cathode and anode ('rock chairs') has achieved theoretical value due to the limited theoretical specific capacity of graphite (372 mAh g-1), the current anode of Li-ion batteries. | |
| Compared to graphite, metallic lithium anode with an extremely high theoretical specific capacity (3860 mAh g-1) and the lowest reduction potential (-3.04 V vs standard hydrogen electrode) has been strongly reconsidered as a promising next-generation anode, which can effectively improve the energy density of rechargeable batteries. | |
| The research of metallic Li as anode originates from 1970s, however, the seemingly inevitable safety problem hinders the practical application of Li batteries. | |
| "During the repeated charge and discharge cycles, the deposited Li cannot recover the initially uniform morphology but form branched or tree-like structure of Li deposits on the anode surface, which pierces the separator easily and generates much dead Li, causing the short circuit and even explosion of batteries," says Dr. Qiang Zhang, a faculty at Department of Chemical Engineering, Tsinghua University, China. "Therefore, safety and uniform deposits of Li ion are critical issues for promoting the practical application of metallic Li as anode for post Li-ion batteries, including rechargeable Li–S, Li–air batteries, and even Li metal batteries which utilize intercalation compounds as cathodes." | |
 |
|
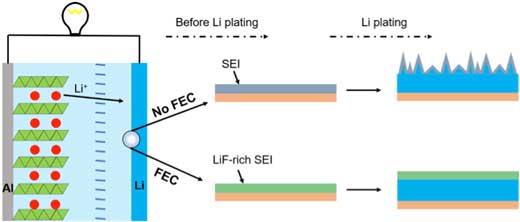
| Fig. 1. Fluoroethylene carbonate (FEC) additives are used to form a LiF-rich solid electrolyte interphase (SEI). The FEC-induced SEI layer is compact and stable, and thus beneficial to obtain a uniform morphology of Li deposits in a working Li metal batteries. | |
| "When metallic Li contacts with the electrolyte, solid electrolyte interphase (SEI) is generated on the surface of Li anode, of which the structure and components play a critical role on the uniform Li deposits," says Qiang. "Electrolyte additives is an effective method to improve the stable and cycling performance of Li-ion batteries in industrial manufacture. Therefore, electrolyte additives may be incorporated into the liquid electrolyte to adjust the components of SEI in situ for uniform Li deposits, which can be also extended easily." | |
| Inspired by this concept, Xue-Qiang Zhang, a graduate student in Qiang’s research group, successfully demonstrated a facile but effective regulation strategy to render uniform Li deposits by incorporating fluoroethylene carbonate (FEC) additives (see Fig. 1). | |
| "LiF has been proved to be an effective component for uniform Li deposits according to previous reports," says Xue-Qiang. "However, the methods in those reports are either complex or toxic, which are not suitable to industrial manufacture. So, we design an effective method to render the uniform Li deposits and circumvent those trouble at the same time. FEC additive can generate more LiF on the surface of Li anode forming the LiF-rich SEI to improve the deposits of Li, the whole process being nontoxic and easy to handle." | |
| "Moreover, we firstly investigated the SEI formation induced by FEC additive by first-principles calculation, which guides the whole experiment and confirms the results effectively." | |
| "The use of polysulfides as additive is another route to obtain stable SEI," notes Chen-Zi Zhao, another graduate student in Qiang’s research group. "We have fabricated a family of electrolytes contain Li2S8, Li2S7, to Li2S, and found the addition of Li2S5 is very effective to form a double layered SEI in a working Li metal cell." | |
| The rational combination of Li2S5 with LiNO3 and LiTFSI can render Li metal anode with dendrite-free morphology, improved Coulombic efficiency, suppressed polarization, and prolonged lifespan (see Fig. 2). | |
 |
|
| Fig. 2. The use of polysulfide containing electrolyte to build protective SEI layer on Li metal anode. | |
| This research group also demonstrates that the FEC and polysulfides induced SEI layer is compact and stable, thus beneficial to obtain a uniform morphology of Li deposits. | |
| This uniform and dendrite-free morphology renders a significantly improved Coulombic efficiency of 98% within 100 cycles in a Li | Cu half-cell. When the FEC-protected Li metal anode matched high loading LiNi0.5Co0.2Mn0.3O2 (NMC) cathode (12 mg cm-2), the FEC-protected Li metal anode guarantees the efficient operation of LMB with long cycle life. | |
| "The strategy of Li metal battery with Li metal anode and intercalation-type NMC cathode affords an emerging energy storage system with very high energy density," says Qiang. "The in situ generated stable LiF-rich SEI strategy is also expected to be applied in other metal batteries (e.g. Li-S, Li-air batteries) with dendrite-free growth and very high Coulombic efficiency." | |
| This research group also compared the gap between the coin cell and pouch cell and found the failure of Li metal anode was a main reason for the capacity decay and short cycling life. More attentions should be concentrated on efficiently protecting Li metal anode to achieve large capacity and safe Li metal cells with high energy density. | |
References |
|
| Zhang X-Q, Cheng X-B, Chen X, Yan C, Zhang Q. Fluoroethylene Carbonate Additives to Render Uniform Li Deposits in Lithium Metal Batteries. Adv. Funct. Mater. 2017, 27, 1605989. | |
| Zhao CZ, Cheng XB, Zhang R, Peng HJ, Huang JQ, Ran R, Huang ZH, Wei F, Zhang Q, Li2S5-based Ternary-Salt Electrolyte for Robust Lithium Metal Anode. Energy Storage Materials 2016, 3, 77-84. | |
| Cheng XB, Yan C, Huang JQ, Li P, Zhu L, Zhao LD, Zhang YY, Zhu WC, Yang ST, Zhang Q. The gap between long lifespan Li-S coin and pouch cells: The importance of lithium metal anode protection. Energy Storage Materials 2017, 6, 18-25. | |
|
Provided by Tsinghua University as a Nanowerk exclusive
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
