| Posted: May 03, 2017 | |
Spermbots - microrobotics meets sperm cells |
|
| (Nanowerk Spotlight) Bio-hybrid microswimmers are devices that move on a small scale and are a combination of biological and artificial components. The biological component can offer a biocompatible propulsion source, the cargo itself and additional features such as sensing. | |
| The artificial part is crucial for remotely controlling the direction they are swimming, e.g. by external magnetic fields. These microswimmers are promising tools for the performance of tasks at the small scale, for instance, making medical operations less invasive. | |
| A progress report recently published in Advanced Materials ("Spermatozoa as Functional Components of Robotic Microswimmers") describes how spermatozoa can be useful parts of microdevices: As biocompatible propulsion source, but also entailing other functionalities such as their natural destiny for fertilization, their ability to respond to stimuli, or their ability to take up drugs open up fascinating new applications. | |
| This article was a joint effort by researchers from the Micro- and Nanobiomedical Engineering Group (MNBE) of the IFW Dresden, Germany, and Dr. Jens Elgeti, biophysicist from the Forschungszentrum Juelich, Germany, who contributed with theoretical considerations on spermatozoa as integrated parts of robotic microswimmers. | |
| The authors recently demonstrated methods for the remote controlled single sperm cell capture, transport and delivery. These are the first examples of using sperm cells as robotic components; the so-called “spermbots” are also systems that enable biophysical studies, e.g. of sperm motion in confinement. | |
| The article describes the different fabrication routes to create spermbots which are characterized by an optimal interaction between the artificial components and the sperm cells. | |
| Spermbots are unique ways for for remote-controlled single sperm cell delivery. On the one hand, motile sperm cells can enter tiny magnetic microtubes and propel them forward (tubular spermbots) and are guided by external magnets. | |
| On the other hand, immotile, but otherwise intact sperm cells can be captured and driven by microhelices which are actuated by external rotating magnetic fields (helical spermbots). Both these microstructures are promising tools for novel assisted reproduction techniques. They target specifically male infertility cases in which only few sperm cells are present (oligospermia) or the sperm cells are immotile (asthenospermia). | |
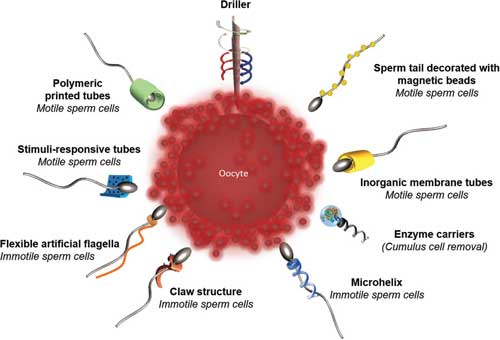 |
|
| Novel microswimmer designs for targeting fertilization. From the top and clockwise: magnetically actuated drillers for penetrating the cumulus layer of the oocyte; the sperm tail decorated with magnetic beads for magnetic actuation of the tail. Rolled up microtubes made of inorganic nanomembrane for the guidance of motile sperm cells; magnetic helical enzyme carriers for the cumulus cell removal by enzymes (e.g., hyaluronidase); artificial flagella with multiple windings and a claw-shaped sperm carrier for the delivery of immotile sperm cells; artificial flexible flagella for transport of immotile spermatozoa; stimuli-responsive polymeric microtubes for the delivery of motile spermatozoa; and polymeric printed microtubes for the guidance of motile sperm cells. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The article summarizes for the first time the advances in the integration of single sperm cells in microrobotic devices and illustrates how wide-ranging the potential of these reproductive cells is. | |
| These cells are very promising as propulsion sources in physiological conditions and thereby enable the actuation of robotic devices under these conditions. | |
| The progress report also provides insight into the current challenges and influencing factors on the performance of the sperm-driven microswimmers. | |
| The challenges ahead include the in vivo imaging of the microstructures which is a requisite to implementing the sperm-driven structures for in vivo applications. | |
| Furthermore, the controlled and high-throughput coupling between the sperm cells and the microstructures remains a major challenge. Sperm selection is an important issue which has to ensure the best spermatozoa are used for fertilization. The removal or degradation of the synthetic components after use appear as some of the crucial requirements for in vivo applications. | |
| The authors also discuss the potential applications in other biomedical scenarios such as drug delivery for treatment of diseases in the female reproductive tract. | |
| In the near future, the researchers will explore the drug delivery potential of spermbots and also the ability to perform successful remote-controlled fertilization. | |
| They will also improve the design of the spermbots to improve their performance including adaptation to different environments, higher velocities, better control and more functionalities. | |
| By Dr. Veronika Magdanz, IFW Dresden | |
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
