| Posted: Oct 03, 2017 | |
Electrode layer prepared from activated carbon (from tea leaves) enhances fuel cell performance by 33% |
|
| (Nanowerk Spotlight) Fuel cells are considered as promising energy technology of the future, as they aid in production of sustainable energy using hydrocarbons as fuels, such as methanol, ethanol, acetone etc by simple oxidation-reduction reaction mechanism. Hence find their use in laptop chargers, military applications, where the access to electricity is difficult. | |
| However the wider spectrum of commercial perspective of methanol systems is greatly hindered by the mass transport phenomenon occurring in the electrode area of a fuel cell. | |
| In a fuel cell, methanol undergoes oxidation on anode side, liberating protons and electrons. Electrons travel through external circuit and protons travel through membrane and reach the cathode side. On the cathode side, air undergoes reduction on cathode side, utilizing electrons and protons from anode reaction to form water. | |
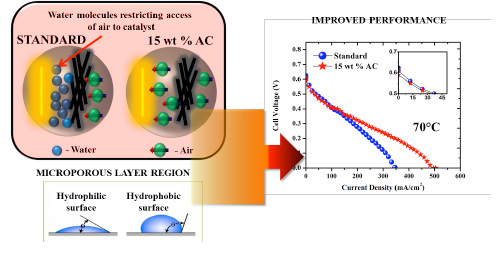
| This water, where the rate of formation is high at high current density regions covers the catalyst sites and prevents the incoming the air stream from further accession. This phenomenon – called ‘flooding’ – limits the operation of fuel cells at high current density regions. This is mitigated by optimising pore characteristics of carbon materials used in the gas diffusion layer of membrane electrode assembly (MEA) of a fuel cell. | |
| MEA of a fuel cell consists of gas diffusion layer, catalyst layer and the membrane, all sandwiched together, with each layer playing a prominent role. The gas diffusion layer aids in effective reactant transport, catalyst layer in oxidation/reduction reaction and the membrane layer in transport of protons from anode to the cathode. | |
 |
|
| Figure 1: Schematic illustration of impact of using activated carbon (AC) in the gas diffusion layer comparable to standard (STD) MEA. (Image: University of Manchester) | |
| The mass transport limitation is addressed by working on the pore structure of carbon materials. Several carbon blacks: Vulcan XC-72, Ketjen Black, Acetylene Black Asbury 850, Denka Black and Mogul L are used widely. In addition to the performance limitation, the precursor for these carbon materials comes from the treatment of hydrocarbons at high temperature and pressure conditions. | |
| In this work, reported in Electrochimica Acta ("Enhanced performance based on a hybrid cathode backing layer using a biomass derived activated carbon framework for methanol fuel cells"), Manchester University chemical engineering researchers Prabhuraj Balakrishnan and Stuart M. Holmes prepared an activated carbon framework from tea leaves, obtained from the Black sea region of Tukey (from Ankara University) collaborators. | |
| This framework used in combination with conventional carbon black in the cathode gas diffusion layer, has shown to improve the fuel cell performance by 33% in methanol fuel cell operating conditions. | |
| More specifically, the cell performance underwent drastic changes in the mass transport region of the fuel cell polarization curve (Figure 1), comparable to the standard MEA. This is attributed to the pore structure (75.69% of mesopores and 24.31% of micropores) of this framework aiding in enhanced water removal, as a result more air molecules react with the platinum catalyst sites finally improving the fuel cell performance. | |
| On increasing the composition of this activated carbon framework in the gas diffusion layer, the performance declined due to the poor electrical conductivity of the carbon. This shows that if the electrical conductivity of this activated carbon framework could be improved, this would decrease the reliance on conventional carbon blacks which currently suffers from source material (hydrocarbon) availability in addition to improving performance. | |
|
Provided by The University of Manchester as a Nanowerk exclusive
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
