| Posted: Mar 02, 2018 | |
Fluorinated phosphorene: a new way of solving instability problems in phosphorene |
|
| (Nanowerk Spotlight) Recently, a new member has been introduced to the family of two-dimensional (2D) materials: phosphorene. Phosphorene has attracted much attention because of its tunable direct band gap (0.3∼2.0 eV) and superior carrier mobility (∼103 cm2/Vs), but the fly in the ointment is that phosphorene is subjected to oxidation and degradation under ambient conditions. | |
| In a recent article published in Small ("Fluorinated phosphorene: electrochemical synthesis, atomistic fluorination, and enhanced stability"), Professor Han Zhang, director of Shenzhen Engineering Laboratory of Phosphorene and Optoelectronics at Shenzhen University, reported a facile pathway in obtaining and stabilizing phosphorene through a one-step, ionic liquid-assisted electrochemical exfoliation and synchronous fluorination process. | |
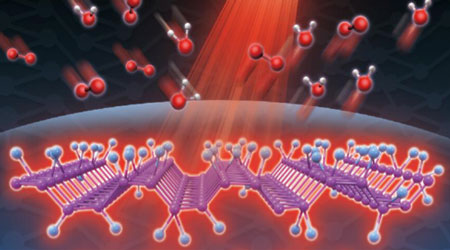
| This strategy enabled a novel phosphorene derivative to be discovered – fluorinated phosphorene (FP). The highly electronegative fluorine adatoms in FP act as a protective umbrella to prevent FP from the attack from ambient O2 and H2O, endowing FP robust ambient stability. | |
| The synthesized FP, with a photo-thermal conversion efficiency of 27.4%, exhibits air-stable photo-thermal properties, indicating that FP could be further developed with a good photo-sensitizer with enhanced stability for tumor or cancer treatments. | |
 |
|
| Figure 1. This illustration shows that phosphorene can withstand the attack from O2 and H2O through the introduction of fluorine adatoms. The fluorinated phosphorene can also generate considerable heat under near-infrared light illumination. (Image: Dr. Xian Tang, Shenzhen University) | |
| Stabilization strategies for phosphorene proposed previously involved either post-treatment of phosphorene or advance synthesis of functional groups, which might increase the technical complexity as well as the possibility of in-process oxidation or degradation. | |
| Preferably, the proposed synchronous fluorination strategy for phosphorene stabilization is realized during its synthesis process, which overcomes the problems encountered in previous stabilization strategies. | |
| Possible atomistic fluorination configurations of FP were revealed by core-level binding energy shift calculations in the Born-Haber cycle in combination with X-ray photoelectron spectroscopy measurements. The results indicate that electrolyte concentration significantly modulates the fluorination configurations. | |
| Taking advantage of versatility and scalability, electrochemical strategies have been applied in many fields such as metal refinement, gas production, and electrode evaluation. In recent years, electrochemical exfoliation, a newly developed synthesis strategy, has become more and more popular for synthesizing 2D materials. | |
| However, the aqueous electrolyte previously used for electrochemical exfoliation of phosphorene inevitably produces oxygen-containing groups and would have a severe impact on the stability of the exfoliated phosphorene. | |
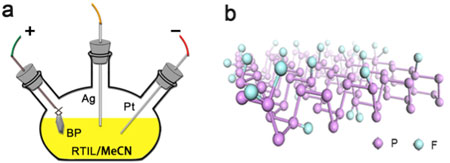
| In this research, ionic liquid, a conductive non-volatile and eco-friendly room temperature molten, is introduced as a non-aqueous electrolyte for the electrochemical exfoliation and synchronous fluorination of phosphorene. This ingeniously solves the problem encountered in the aqueous electrochemical exfoliation by the decoration of ionic liquid-electrolyzed F- ions on phosphorene. | |
 |
|
| Figure 2. (a) Schematics of (a) electrochemical synthesis setup and (b) atomic structure of fluorinated phosphorene. (Image: Dr. Xian Tang, Shenzhen University) | |
| As more and more 2D materials are emerging that potentially suffer from environmental vulnerabilites like phosphorene, enough attention should be paid to this issue and advanced stabilization strategies should be developed accordingly. | |
| The fluorination stabilization strategy is a bold attempt and can be easily extended to other environmentally sensitive 2D materials. | |
|
Provided by Shenzhen Engineering Laboratory of Phosphorene and Optoelectronics, Shenzhen University, as a Nanowerk exclusive
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
