| Nov 19, 2019 | |
From origin of life to materials genomics |
|
| (Nanowerk Spotlight) The origin of early life and the basic building blocks such as DNA and RNA have been hypothesized to have resulted due to accumulation of precursors within hydrothermal vents. The thermal gradients result in the concentration of prebiotic molecules with the vents acting as reactors. | |
| In this work (Matter, "Accumulation-Driven Unified Spatiotemporal Synthesis and Structuring of Immiscible Metallic Nanoalloys"), we translate the above concept of accumulation-mediated concentration to the micro- and nanoscale for intensified materials synthesis and genomics. | |
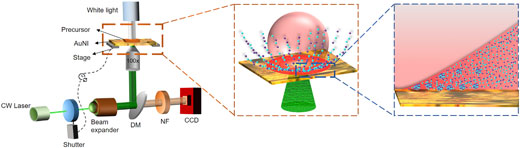
| We utilize a laser-induced micro bubble trap (MBT), which acts as an accumulator-cum-reactor, similar to the hydrothermal vents where supposedly life originated. Our strategy, termed as unified spatiotemporal synthesis and structuring (US3), combines the conventionally discrete aspects of synthesis and patterning. | |
| This novel approach of in situ material synthesis and additive manufacturing can result in reduced material contamination, corrosion and wastage. | |
 |
|
| Schematic Demonstrating the Optical Setup and Ion-Accumulation Aspect of the US3 Strategy. The laser-induced MBT can lead to highly localized supersaturations of precursor ions near the substrate, enabling the simultaneous synthesis and structuring of immiscible alloys. (Reprinted with permission by Cell Press) (click on image to enlarge) | |
| We apply our accumulation-driven technique toward the synthesis and simultaneous structuring of immiscible nanoalloys. | |
| Bimetallic nanoalloys have been found to exhibit unique and significantly enhanced optical, electrical and catalytic characteristics, which are distinct from the pure metals. | |
| Like the creation of complex multicomponent biological building blocks, the blending of multiple elements is an attractive strategy to tune the properties of inorganic materials via atomic ordering, size and composition. | |
| As a case in point, we synthesize and print rhodium-gold (RhAu) nanoalloys in a one-shot process at the MBT and ∼120 °C. RhAu nanoalloys are challenging systems to synthesize, with these alloys predicted to have no stable regions below ∼1850 °C and 1.0 atm. | |
| Using a laser-induced micro bubble trap to synthesize and print rhodium-gold nanoalloys with interline spacings from 2 to 8 µm. | |
| In our technique, we can accumulate precursor ions at ultra-high supersaturation within millisecond timeframes at the MBT. This enables a 3-order reduction in synthesis time, from seconds/minutes for current protocols toward millisecond timeframe via US3. | |
| We've also utilized stochastic random was simulations to trace the motion of individual ions and demonstrate the ultra-fast accumulation process. Upon printing, we demonstrate the catalytic activity of the fabricated RhAu patterns using reduction of p-nitrophenol to p-aminophenol by NaBH4 as a model study and show superior catalytic activity of the nanoalloy. | |
| Our new strategy provides a way to fabricate and characterize a variety of new materials for superior functionalities. In combination with a microfluidic device, US3 platform can enable high-throughput development and screening of various multi-element nanomaterials with tailored catalytic, optical, electronic and magnetic functions. | |
|
By Bharath Bangalore Rajeeva, PhD, Materials Science and Engineering, Texas Materials Institute, The University of Texas at Austin
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
