| Jul 22, 2020 | |
A new breakthrough in lithium-silicon batteries |
|
| (Nanowerk Spotlight) Lithium-ion batteries are ubiquitous in today’s electronic devices such as smart phones and laptops. Even NASA uses rechargeable batteries in many missions to provide electrical power for survival during eclipse periods on solar-powered missions (e.g., low-earth orbiting satellites, international space station), and as a power source for astronaut suits for enabling them to perform extra vehicular activities. Indeed, lithium-ion batteries have also been used in Mars landers and rovers. | |
| Unfortunately, the state-of-the-art lithium-ion batteries are heavy and bulky. Graphite, the soft and flaky material in pencil lead, has long been a key component in lithium-ion batteries. | |
| At the microscopic scale, graphite consists of multiple layers of carbon stacked on top of one another. In a traditional lithium-ion battery, lithium ions zip in and out of the vacant gallery spaces between these layers in graphite. Although graphite is a very efficient anode, it cannot store much lithium. | |
| Therefore, it takes a large number of graphite-based cells to provide the needed energy for space missions leading to heavier battery packs that must be transported into space. This low capacity of graphite electrodes is also disadvantageous for the use of lithium-ion batteries in electric cars or other battery powered tools. | |
| “Rechargeable batteries are important sources for providing electrical power for survival during eclipse periods on solar-powered missions,” Ramakrishna Podila, an Assistant Professor at Clemson University, tells Nanowerk. “For example, the batteries in the ISS are charged via the solar arrays during the approximately 45-minute period when the arrays are in direct sunlight each orbit and are discharged as they power the station’s loads during the other 45-minute period of darkness per orbit.” | |
| A transition to lightweight lithium-ion batteries is direly needed. The integration of graphite-based batteries into NASA missions or electric cars has been extremely limited because of their higher weight. | |
 |
|
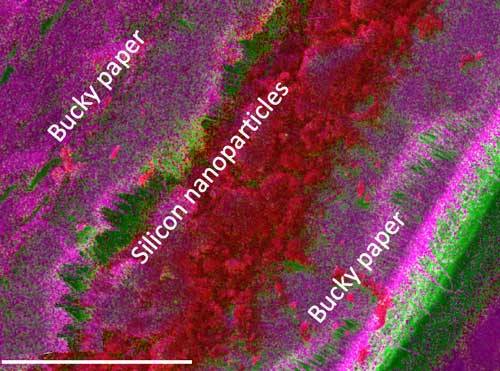
| A cross-sectional elemental map showing the microscopic structure of the electrode with silicon nanoparticle (scale bar is 100 µm). (Image courtesy of the researchers) | |
| Silicon has ten times higher capacity than graphite. Replacing graphite with silicon could lead to lighter and safer batteries. Although silicon can take on more lithium than graphite, it tends to expand about 300 percent in volume, causing the anode to become electrically insulating and break apart. This limits the cycle life of silicon-based lithium batteries to less than 100 cycles. | |
| Reporting their findings from a project funded by NASA, scientists from Clemson Nanomaterials Institute have recently discovered a novel 'sandwiched' silicon electrode structure that can withstand 500 cycles and deliver capacities three times larger than graphite. | |
| Their results are published in ACS Applied Materials and Interfaces ("Three-dimensional Si anodes with fast diffusion, high capacity, high rate capability, and long cycle life"). | |
| “We used freestanding sheets made of carbon nanotubes, called bucky papers, for sandwiching silicon nanoparticles. These nanotubes form a quasi-three-dimensional structure and hold silicon nanoparticles together even after 100 cycles and mitigate electrical resistance arising from breaking of particles,” explains Shailendra Chiluwal, a graduate student at CNI and the first-author on the study. | |
| The sandwiched silicon anode was able to withstand discharging rates as high as 4C (At 4C rate, the battery charges fully within 15 minutes). They also show a stable capacity as high as 1650 mAh/g up to 500 cycles. The team showed that the diffusion time constant, for Li ion transport in and out of the anode, is increased by 150 times by using bucky papers. | |
| “Silicon as the anode in a lithium-ion battery represents the 'holy grail' for researchers in this field,” remarks Prof. Apparao M. Rao, who is director of the Clemson Nanomaterials Institute and the principal investigator on the NASA grant. “Carbon nanotubes in bucky papers are permeable to lithium ions unlike traditional current collectors such as copper. This allows bucky papers to enable faster ion transport” | |
| The team used extensive electrochemical impedance spectroscopy and ex-situ energy dispersive x-ray analysis to show that carbon nanotubes provide electrical connections between silicon nanoparticles even after 100 cycles unlike traditional silicon electrodes. | |
| "Our next goal is to collaborate with industrial partners to translate this lab-based technology to the marketplace," notes Podila, corresponding author of the study and a co-investigator on the NASA grant. "We are thankful to the NASA and South Carolina EPSCoR for granting an award to undertake such projects which would have lasting impact on space missions and the global energy landscape." | |
|
Provided by Clemson Nanomaterials Institute as a Nanowerk exclusive
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
