| Posted: Aug 06, 2008 | |
Imaging a single quantum dot when it is dark |
|
| (Nanowerk Spotlight) More than half a century ago, Erwin Schrödinger, nobel laureate in physics, claimed that it is "impossible to carry out experiments on single molecules or atoms". Today, the detection, tracking and study of single molecules and atoms has become an omnipresent tool in biology, chemistry and physics alike. For example, sequencing DNA one base pair (or letter) at a time currently provides the most likely solution to fulfill the quest for a $1,000 human genome. | |
| Nevertheless, observation of a single molecule, especially with standard light microscopes requires a good deal of laboratory skills. This is mostly due to the fact that a single molecule only gives a miniscule amount of detectable signal. In fact, people using light as a probe have relied exclusively on the use of fluorescence, the emission of lower energy light following absorption of radiation at a certain energy. In this scheme, the signal from the molecule of interest can be easily separated from residual excitation light or background fluorescence simply by filtering the detected light spectrally and only detecting the color that is emitted by the molecule. In this way, it is possible to suppress unwanted signals from the billions of other molecules that are in the vicinity of the molecule of interest. | |
| As powerful as this approach has been, it also has one major limitation: it is only possible to study molecules that are highly fluorescent, i.e. emit lower energy light with high efficiency. Examples include mostly synthetic molecules such as dyes but also some natural ones like the green fluorescent protein (GFP). Unfortunately, only few hundreds of molecules are good enough emitters such that they can be detected at the single molecule level. | |
| To avoid this limitation, many attempts have been made to detect the light absorbed rather than that emitted by molecules because a larger number of molecules absorb than emit efficiently. | |
| Scientists from the ETH Zurich in the group of Vahid Sandoghdar have recently demonstrated a major step towards the detection and study of single molecules in absorption. In their proof-of-principle study ("Imaging a Single Quantum Dot When It Is Dark"), they report the extinction of laser light by single quantum dots, molecular-sized balls of semiconductor material. In addition, they were able to show how the magnitude of the absorption of the dots depends on their well-being, i.e. their so-called photophysical state giving insight into their basic photophysics. | |
| The ETH group had shown in a series of experiments that it is possible to detect single nonfluorescent nanoparticles using light microscopy, whereas for years this was considered to belong to the realm of electron microscopy. | |
 |
|
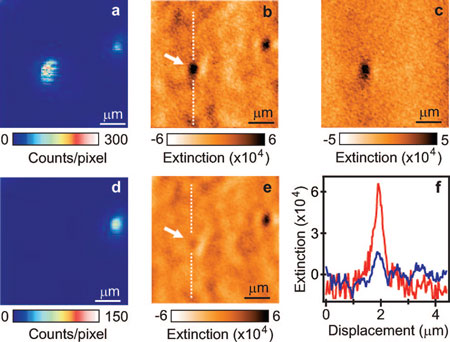
| Fluorescence (a) and extinction (b) images of two individual D2 quantum dots. The marked dot was the same as the one studied in Figure 2. (c) Difference between images (b) and (e). (d,e) Images obtained after illuminating the marked nanocrystal quantum dot with 20 kW/cm2 for several minutes. (f) Cross section of the quantum dot before (red) and after (blue) photobleaching. (Reprinted with permission from American Chemical Society) | |
| In their earlier works, they showed that by interfering a laser beam with the tiny amount of light that is scattered (i.e. light scattered at the same wavelength/color as the incident laser) by a nano-object, it is possible to obtain a detectable contrast in case of single gold particles as small as 5 nm in diameter ("Detection and Spectroscopy of Gold Nanoparticles Using Supercontinuum White Light Confocal Microscopy"), individual microtubules ("Interferometric optical detection and tracking of very small gold nanoparticles at a water-glass interface"), and 45 nm large single viruses ("Label-Free Optical Detection and Tracking of Single Virions Bound to Their Receptors in Supported Membrane Bilayers ") without the need for any labeling. | |
| Down deep, this interferometric technique turns out to be nothing but a way to detect the small amount of light that is extinguished from the incident laser beam due to absorption or scattering. | |
| Aside from reducing the background scattering, the main trick used by the ETH team to reach the sensitivity required for seeing one quantum dot is to optimize the phase between the laser light and the light scattered by the dot. They have succeeded in detecting an absorption of the order of 0.01%. | |
| In their recent report, the ETH scientists have also shown that they can interrogate and image a quantum even when it does not emit, e.g. after it has photobleached or during photoblinking off times. In other words, extinction imaging allows one to observe the system of interest "even when it is turned off". | |
| The results of Sandoghdar and coworkers indicate that it should absolutely be possible to detect the absorption of light by single molecules, which provide signals only several times smaller than the studied dots, and thereby detect and study them irrespective of their abilities as emitters. | |
| Many other fields may benefit hugely from this approach. To bring sensing and detection of (non-fluorescent) bio-molecules as well as trace substances at very small concentrations to the single molecule level has been a long standing task and appears more manageable given these results. In other words, the future is bright for seeing in the dark. | |
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
