| Posted: March 23, 2009 |
The growth of nanotoxicology |
|
(Nanowerk News) The use of engineered nanomaterials in consumer products is expanding - a current report by the Woodrow Wilson International Center for Scholars and the Pew Charitable Trusts identifies more than 800 commercial nanomaterial-containing products, accounting for $147 billion (approximately £104 billion) yearly. The materials show promise in disease treatment or solar power generation. Yet, despite the fact that so many are in commercial use, very little is known about their effects on health.
|
|
As scientists around the world try to fill this information void, nanotoxicology research has grown rapidly and a wide variety of analytical techniques are used to assess biodistribution (tracking where the compounds travel in the body), cellular uptake and both in vivo and in vitro toxicity (Analytical methods to assess nanoparticle toxicity – free access article).
|
|
Nanotoxicity experiments are typically conducted on mice or rats and focus on LD50 (exposure amount resulting in 50 per cent population death), changes to tissues or organs, or changes in blood cell populations and serum. These experiments give valuable information but are often time consuming, expensive and provide relatively little mechanistic information about underlying toxicity causes. There is also an ethical imperative to reduce the large animal numbers used in these studies.
|
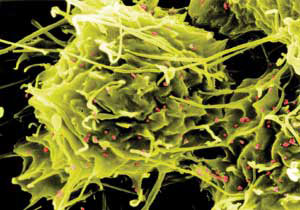 |
| Toxicology studies reveal nanoparticles' uptake in the body, for example, the iron oxide particles (red) on the surface of white blood cells shown here. (Image: Harald Krug, EMPA, St Gallen, Switzerland, and Hellmuth Zöltzer, University of Kassel, Germany)
|
|
In vitro assessment may be a better alternative. It can provide inexpensive and rapid nanomaterial interaction analysis on the cellular level. Material uptake and location can be assessed using electron microscopy, fluorescent confocal microscopy or elemental analysis. On their own, these techniques have limitations so are best used in concert to get a good representation. In vitro assessment often relies on using bulk tissue samples from immortalised cell lines and toxicity biomarker probe molecules. In vivo toxicity is difficult to predict from the results - some nanomaterial classes (such as carbon nanotubes) interact with probe molecules directly, providing misleading results.
|
|
Scientists have developed new nanomaterial distribution and toxicity methods to tackle these challenges, but further methodological developments are needed. These include toxicity analysis techniques to discriminate individual cellular function within mixed culture environments, label-free dynamic nanoparticle uptake analysis, nanoparticle surface characterisation within complex biological environments and point-of-source nanoparticle exposure analysis for workers.
|
|
Also, the worldwide nanotechnology community would benefit greatly from a set of standard toxicity screening protocols for engineered nanomaterials. This would allow scientists to develop safe nanotechnologies and would lessen public fear regarding exposure to nanomaterials, ultimately helping to unlock the full potential of these exciting materials.
|

