| Oct 24, 2012 |
New self-assembled nanoparticles may soon make biopsies obsolete
|
|
(Nanowerk News) Early detection of soft-tissue diseases, such as breast cancer, typically requires invasive biopsies. Now, a new self-assembled nanoparticle developed by Bin Liu at the A*STAR Institute of Materials Research and Engineering and co-workers may soon make biopsies obsolete. The team’s material significantly enhances the safety of two-photon microscopy (TPM) — a technique that uses fluorescent probes to generate three-dimensional pictures of cancer cell structures in living tissue ("Glycosylated Star-Shaped Conjugated Oligomers for Targeted Two-Photon Fluorescence Imaging").
|
|
Although TPM provides deep access to cell tissue without significant photo-damage, finding suitable substances to act as light-emitting probes is challenging. ‘Quantum dots’ made from nanoscale aggregates of elements such as cadmium and selenium are excellent cell-structure illuminators, thanks to their bright and stable fluorescence. However, their inherent toxicity restricts many possible biological applications.
|
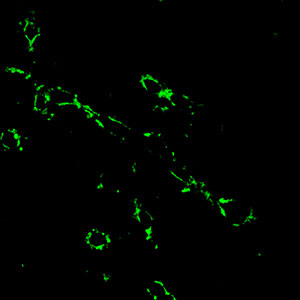 |
| A new fluorescent glucose-amine probe can make identification of cancer cells (green) using two-photon microscopy easier and safer.
|
|
Liu and her team therefore turned to conjugated organic molecules to produce less toxic dyes for TPM. While such small organic molecules are normally unable to absorb sufficient amounts of laser light to initiate fluorescence imaging, the team resolved this problem by synthesizing a star-shaped material known as a dendrimer. Consisting of a central triphenyl amine core and three ‘arms’ made from extended conjugated chains, this unique geometry can induce much larger cross sections that can absorb two-photons better than isolated fluorescent dyes.
|
|
To ensure biocompatibility between the star-shaped dendrimer and cell tissue, the researchers had to employ a chemical trick. Inspired by the versatile binding behavior of chitosan, a natural polysaccharide, the team used a mild bromide–thiol reaction to attach several glucose–amine sugar rings to the dendrimer’s arms. According to Liu, this process lowered the cytotoxicity of the dye and enabled them to functionalize it with folic acid ligands that target the surfaces of a breast cancer cell line known as MCF-7.
|
|
The team’s experiments showed that the dendritic dye self-assembled into dispersed nanoparticles when submerged in water — a form that increases two-photon-absorption cross sections and provides a high yield of laser-induced fluorescence. When they incubated these nanoparticles into the MCF-7 cells, subsequent TPM imaging revealed a bright fluorescence localized inside the cancer cell cytoplasma (see image). This data indicates that specific binding occurs between the dendritic dye and folate receptors on the MCF-7 surface.
|
|
Cell viabilities close to 100% at dye concentrations used for imaging studies confirms that this strategy is a safe and promising way to increase the use of TPM imaging. “We are keen to expand the current in vitro imaging to in vivo applications,” notes Liu.
|

