The 'dark matter' in the protein universe
 Using bioinformatics methods, an international team of scientists has come one step closer to unveiling the mystery that surrounds the dark proteome.
Using bioinformatics methods, an international team of scientists has come one step closer to unveiling the mystery that surrounds the dark proteome.
Nov 25th, 2015
Read more
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed
 Using bioinformatics methods, an international team of scientists has come one step closer to unveiling the mystery that surrounds the dark proteome.
Using bioinformatics methods, an international team of scientists has come one step closer to unveiling the mystery that surrounds the dark proteome.
Nov 25th, 2015
Read more Against voracious beetles or caterpillars plants protect themselves with cyanide. Certain enzymes release the toxic substance when the plant is chewed. These HNL-called enzymes are also important for industry. Researchers found a new biocatalyst in a fern which outshines all other HNL-type enzymes on the market.
Against voracious beetles or caterpillars plants protect themselves with cyanide. Certain enzymes release the toxic substance when the plant is chewed. These HNL-called enzymes are also important for industry. Researchers found a new biocatalyst in a fern which outshines all other HNL-type enzymes on the market.
Nov 24th, 2015
Read more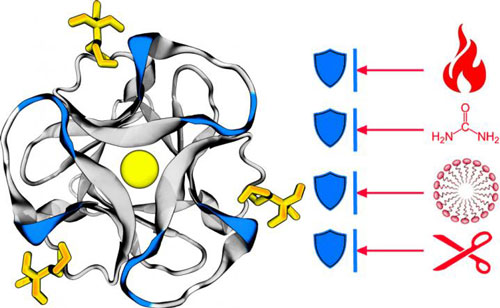 Researchers have discovered a new way to create designer proteins that have the potential to transform biotechnology and personalized medicines.
Researchers have discovered a new way to create designer proteins that have the potential to transform biotechnology and personalized medicines.
Nov 24th, 2015
Read more Bioart ranges from bacterial manipulation to glowing rabbits, cellular sculptures. In the pursuit of creating art, practitioners have generated tools and techniques that have aided researchers, while sometimes crossing into controversy, such as by releasing invasive species into the environment, blurring the lines between art and biology, and challenging scientific thinking.
Bioart ranges from bacterial manipulation to glowing rabbits, cellular sculptures. In the pursuit of creating art, practitioners have generated tools and techniques that have aided researchers, while sometimes crossing into controversy, such as by releasing invasive species into the environment, blurring the lines between art and biology, and challenging scientific thinking.
Nov 23rd, 2015
Read more Scientists have sequenced the complete genetic makeup of a species of ecologically important algae, which may aid in biofuel production.
Scientists have sequenced the complete genetic makeup of a species of ecologically important algae, which may aid in biofuel production.
Nov 19th, 2015
Read more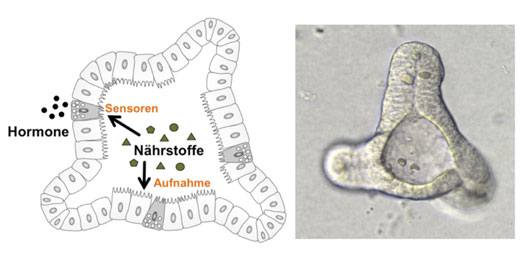 Scientists shown for the first time how artificially grown mini-intestines can be used in nutritional and diabetic research.
Scientists shown for the first time how artificially grown mini-intestines can be used in nutritional and diabetic research.
Nov 19th, 2015
Read more A new injectable biogel is effective in delivering anti-cancer agents directly into cancerous tumours and killing them.
A new injectable biogel is effective in delivering anti-cancer agents directly into cancerous tumours and killing them.
Nov 19th, 2015
Read more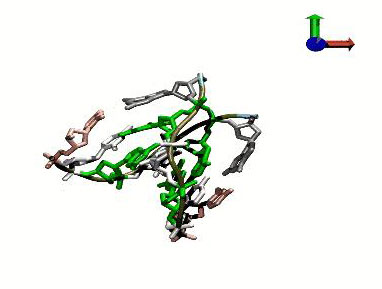 The simulation method allows the study of structural changes in DNA and of the interaction of DNA with proteins and drugs.
The simulation method allows the study of structural changes in DNA and of the interaction of DNA with proteins and drugs.
Nov 17th, 2015
Read more Researchers studying a biological process that enables tiny green algae to grow efficiently have taken the first steps to recreating the mechanism in a more complex plant. Their findings could lead to the breeding of high yield varieties of common crops such as wheat, rice and barley.
Researchers studying a biological process that enables tiny green algae to grow efficiently have taken the first steps to recreating the mechanism in a more complex plant. Their findings could lead to the breeding of high yield varieties of common crops such as wheat, rice and barley.
Nov 16th, 2015
Read moreAbility to introduce or reverse the spread of genetic traits through populations could one day improve pest management and disease control.
Nov 16th, 2015
Read moreScientists have created molecules that mimic and dominate toxic ones secreted by bacteria. The implications for the study of biology and pharmacology are said to be enormous.
Nov 13th, 2015
Read more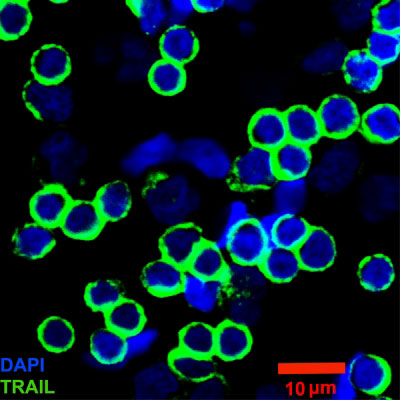 Biomedical engineers have developed specialized white blood cells - dubbed 'super natural killer cells' - that seek out cancer cells in lymph nodes with only one purpose: destroy them. This breakthrough halts the onset of metastasis.
Biomedical engineers have developed specialized white blood cells - dubbed 'super natural killer cells' - that seek out cancer cells in lymph nodes with only one purpose: destroy them. This breakthrough halts the onset of metastasis.
Nov 13th, 2015
Read more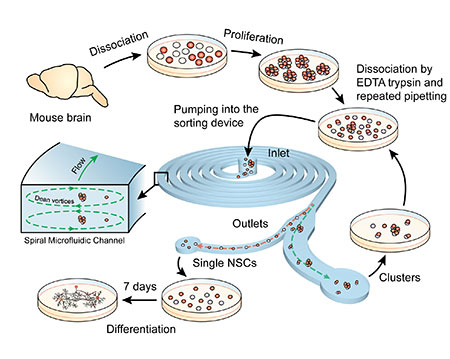 Method safely isolates single stem cells from larger clusters.
Method safely isolates single stem cells from larger clusters.
Nov 9th, 2015
Read more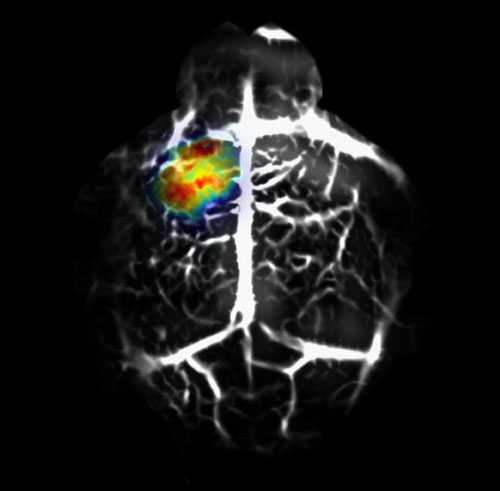 Using a high-tech imaging method, a team of biomedical engineers was able to see early-developing cancer cells deeper in tissue than ever before with the help of a novel protein from a bacterium.
Using a high-tech imaging method, a team of biomedical engineers was able to see early-developing cancer cells deeper in tissue than ever before with the help of a novel protein from a bacterium.
Nov 9th, 2015
Read more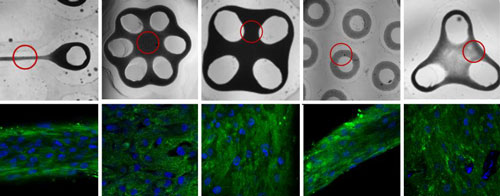 Scientists report culturing cells to make extracellular matrix of two types and five different alignments with the strength found in natural tissue and without using any artificial chemicals that could make it incompatible to implant.
Scientists report culturing cells to make extracellular matrix of two types and five different alignments with the strength found in natural tissue and without using any artificial chemicals that could make it incompatible to implant.
Nov 9th, 2015
Read more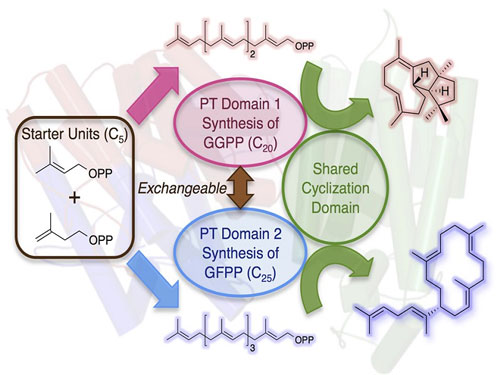 Researchers have discovered and described the function of a unique enzyme involved in the biosynthesis of terpene (terpenoid) natural products, and successfully altered its function by altering its structure so that the enzyme produces another molecule.
Researchers have discovered and described the function of a unique enzyme involved in the biosynthesis of terpene (terpenoid) natural products, and successfully altered its function by altering its structure so that the enzyme produces another molecule.
Nov 9th, 2015
Read more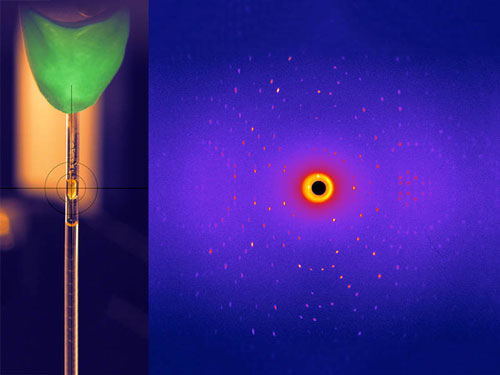 Usually, harvesting energy and raw materials from plants requires many process steps and aggressive chemicals. To make these processes more efficient and resource saving, researchers are looking for suitable enzymes. Using neutrons, researchers have now investigated the reaction mechanism of an important class of enzymes: the glycosidases.
Usually, harvesting energy and raw materials from plants requires many process steps and aggressive chemicals. To make these processes more efficient and resource saving, researchers are looking for suitable enzymes. Using neutrons, researchers have now investigated the reaction mechanism of an important class of enzymes: the glycosidases.
Nov 2nd, 2015
Read more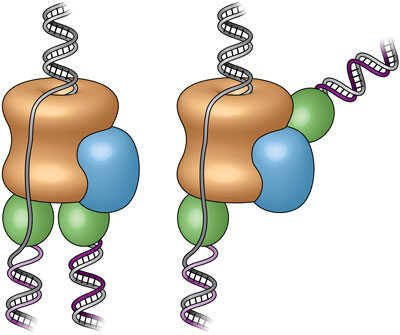 State-of-the-art electron microscope images reveal that structure of DNA-copying protein complex differs from long-held textbook view.
State-of-the-art electron microscope images reveal that structure of DNA-copying protein complex differs from long-held textbook view.
Nov 2nd, 2015
Read more