Growing quantum dots in a regular arrangement
 Researchers have decisively improved the manufacturing process for these tiny semiconductor structures. The quantum dots are generated on a wafer.
Researchers have decisively improved the manufacturing process for these tiny semiconductor structures. The quantum dots are generated on a wafer.
Mar 28th, 2022
Read more
 Researchers have decisively improved the manufacturing process for these tiny semiconductor structures. The quantum dots are generated on a wafer.
Researchers have decisively improved the manufacturing process for these tiny semiconductor structures. The quantum dots are generated on a wafer.
Mar 28th, 2022
Read more A new publiacation reviews the evaluation of metaphotonics induced by artificial intelligence and present a summary of the concepts of machine learning with some specific examples developed and demonstrated for metasystems and metasurfaces.
A new publiacation reviews the evaluation of metaphotonics induced by artificial intelligence and present a summary of the concepts of machine learning with some specific examples developed and demonstrated for metasystems and metasurfaces.
Mar 28th, 2022
Read more Rsearchers show that deep learning can be used to simulate the postfabrication appearance of structures manufactured by FIB milling in the 2D projection of a scanning electron microscope image, as a very good indicator of process accuracy and quality.
Rsearchers show that deep learning can be used to simulate the postfabrication appearance of structures manufactured by FIB milling in the 2D projection of a scanning electron microscope image, as a very good indicator of process accuracy and quality.
Mar 26th, 2022
Read more A team of scientists has developed a so-called 'digital twin' for X-ray experiments and used it to study the production process of novel nanofibers.
A team of scientists has developed a so-called 'digital twin' for X-ray experiments and used it to study the production process of novel nanofibers.
Mar 26th, 2022
Read more The traditional trial-and-error method in material research cannot meet the growing demand of various high-performance materials, and developing effective paradigm of material science is extremely urgent. Researchers propose a new research paradigm for material studies based on the 'functional motif' concept.
The traditional trial-and-error method in material research cannot meet the growing demand of various high-performance materials, and developing effective paradigm of material science is extremely urgent. Researchers propose a new research paradigm for material studies based on the 'functional motif' concept.
Mar 26th, 2022
Read more Study leverages one of the most powerful magnets on Earth to probe a new model of a mysterious metal.
Study leverages one of the most powerful magnets on Earth to probe a new model of a mysterious metal.
Mar 26th, 2022
Read more Semiconductor electronics is getting faster and faster - but at some point, physics no longer permits any increase. The shortest possible time scale of optoelectronic phenomena has now been investigated.
Semiconductor electronics is getting faster and faster - but at some point, physics no longer permits any increase. The shortest possible time scale of optoelectronic phenomena has now been investigated.
Mar 25th, 2022
Read more Scientists use gate set tomography to discover and validate a silicon qubit breakthrough.
Scientists use gate set tomography to discover and validate a silicon qubit breakthrough.
Mar 25th, 2022
Read more Solid-state batteries have long promised higher energy density, faster charging and greater range for electric vehicles than the current generation of lithium-ion batteries, but have suffered from a variety of design obstacles. A novel cathode design however could be a game-changer.
Solid-state batteries have long promised higher energy density, faster charging and greater range for electric vehicles than the current generation of lithium-ion batteries, but have suffered from a variety of design obstacles. A novel cathode design however could be a game-changer.
Mar 25th, 2022
Read more New composite film brings down the heat index or 'felt air temperature' by about 40%.
New composite film brings down the heat index or 'felt air temperature' by about 40%.
Mar 25th, 2022
Read more The maximum speed of signal transmission in microchips is about one petaherz (one million gigahertz), which is about 100,000 times faster than current transistors.
The maximum speed of signal transmission in microchips is about one petaherz (one million gigahertz), which is about 100,000 times faster than current transistors.
Mar 25th, 2022
Read more Researchers have developed shape-programmable 3D microfluidic structures, which are assembled from a bilayer of channel-embedded PDMS and shape-memory polymers via compressive buckling.
Researchers have developed shape-programmable 3D microfluidic structures, which are assembled from a bilayer of channel-embedded PDMS and shape-memory polymers via compressive buckling.
Mar 25th, 2022
Read more A new study introduces a breakthrough in making nanocrystals function together electronically. The research may open the doors to future devices with new abilities.
A new study introduces a breakthrough in making nanocrystals function together electronically. The research may open the doors to future devices with new abilities.
Mar 25th, 2022
Read more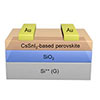 Researchers develop the world's highest performing p-type transistor using perovskite. Solution-processed metal halide perovskite transistors can now be printed.
Researchers develop the world's highest performing p-type transistor using perovskite. Solution-processed metal halide perovskite transistors can now be printed.
Mar 25th, 2022
Read more Realization of stretchable, adhesive, and mechanically deformable batteries that effectively transfer ions. Every component was designed to be stretchable to enable printing on clothing and use in wearable devices.
Realization of stretchable, adhesive, and mechanically deformable batteries that effectively transfer ions. Every component was designed to be stretchable to enable printing on clothing and use in wearable devices.
Mar 25th, 2022
Read more Newly discovered Fermi arcs that can be controlled through magnetism could be the future of electronics based on electron spins.
Newly discovered Fermi arcs that can be controlled through magnetism could be the future of electronics based on electron spins.
Mar 25th, 2022
Read more