 Researchers have found that certain organic semiconducting materials can transport spin faster than they conduct charge, a phenomenon which could eventually power faster, more energy-efficient computers.
Researchers have found that certain organic semiconducting materials can transport spin faster than they conduct charge, a phenomenon which could eventually power faster, more energy-efficient computers.
Mar 26th, 2019
Read more
 In recent years, researchers have tried to capture the electrical current that bacteria generate through their own metabolism. So far, however, the transfer of the current from the bacteria to a receiving electrode has not been efficient at all. Now, they have achieved a slightly more efficient transfer of electrical current.
In recent years, researchers have tried to capture the electrical current that bacteria generate through their own metabolism. So far, however, the transfer of the current from the bacteria to a receiving electrode has not been efficient at all. Now, they have achieved a slightly more efficient transfer of electrical current.
Mar 26th, 2019
Read more
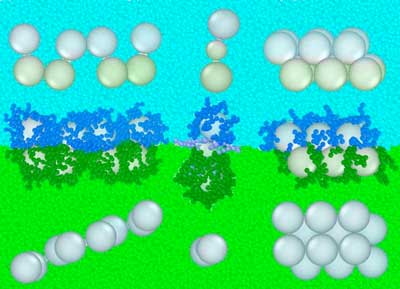 Theoretical approach for assembling nanoparticles uses an idea similar to separated vinaigrettes.
Theoretical approach for assembling nanoparticles uses an idea similar to separated vinaigrettes.
Mar 26th, 2019
Read more
 Researchers demonstrated high light extraction efficiency of perovskite photonic crystals fabricated by delicate electron-beam lithography.
Researchers demonstrated high light extraction efficiency of perovskite photonic crystals fabricated by delicate electron-beam lithography.
Mar 26th, 2019
Read more
 Molecular nanowires can be used for many applications, from LED lights to medical devices.
Molecular nanowires can be used for many applications, from LED lights to medical devices.
Mar 26th, 2019
Read more
 Tiny nanoclusters of metal atoms - such as gold and silver - have properties, which mean they can be used as semiconductors. The finding opens the door to a wide range of potential new applications, from phone displays and flatter screens to wearable technology.
Tiny nanoclusters of metal atoms - such as gold and silver - have properties, which mean they can be used as semiconductors. The finding opens the door to a wide range of potential new applications, from phone displays and flatter screens to wearable technology.
Mar 26th, 2019
Read more
 Scientists have achieved a billion-fold increase in the electrical conductivity of melanin, that could unleash its potential in safe, sustainable bioelectronics.
Scientists have achieved a billion-fold increase in the electrical conductivity of melanin, that could unleash its potential in safe, sustainable bioelectronics.
Mar 26th, 2019
Read more
 There is concern that pollinating insects are at risk due to nanomaterial exposure. Pollinating insects like bees or bumblebees are potentially exposed to nanomaterials via aerosols, the pollen of contaminated plants and water droplets. Managed pollinators may be additionally exposed in the hives due to direct application of nanomaterials by beekeepers.
There is concern that pollinating insects are at risk due to nanomaterial exposure. Pollinating insects like bees or bumblebees are potentially exposed to nanomaterials via aerosols, the pollen of contaminated plants and water droplets. Managed pollinators may be additionally exposed in the hives due to direct application of nanomaterials by beekeepers.
Mar 26th, 2019
Read more
 Scientists have come up with a systematic method for studying and even predicting gene expression - without using cells. Using their innovative, quantitative approach, they measured important parameters governing gene regulation. This allowed them to design and construct a synthetic biological logic gate, which could one day be used to introduce new functions into cells.
Scientists have come up with a systematic method for studying and even predicting gene expression - without using cells. Using their innovative, quantitative approach, they measured important parameters governing gene regulation. This allowed them to design and construct a synthetic biological logic gate, which could one day be used to introduce new functions into cells.
Mar 25th, 2019
Read more
 Cellulose nanofibers hydrogel has great potential as a cell-encapsulation delivery carrier for sustained release of paracrine factors and for tissue regeneration, with unique versatility for injection, scaffolding, and 3D bioprinting.
Cellulose nanofibers hydrogel has great potential as a cell-encapsulation delivery carrier for sustained release of paracrine factors and for tissue regeneration, with unique versatility for injection, scaffolding, and 3D bioprinting.
Mar 25th, 2019
Read more
 Cellulose soaked in a carefully designed polymer mixture acts as a sensor to measure pressure, temperature and humidity -- at the same time. The measurements are completely independent of each other. The sensor may be highly significant in fields such as robotics, healthcare and security.
Cellulose soaked in a carefully designed polymer mixture acts as a sensor to measure pressure, temperature and humidity -- at the same time. The measurements are completely independent of each other. The sensor may be highly significant in fields such as robotics, healthcare and security.
Mar 25th, 2019
Read more
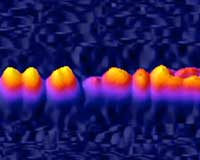 Physicists have shown that shaking ultracold Bose-Einstein condensates can cause them to either divide into uniform segments or shatter into unpredictable splinters, depending on the frequency of the shaking.
Physicists have shown that shaking ultracold Bose-Einstein condensates can cause them to either divide into uniform segments or shatter into unpredictable splinters, depending on the frequency of the shaking.
Mar 25th, 2019
Read more
 A new method allows the quantum state of atomic qubits to be measured with twenty times less error than was previously possible, without losing any atoms.
A new method allows the quantum state of atomic qubits to be measured with twenty times less error than was previously possible, without losing any atoms.
Mar 25th, 2019
Read more
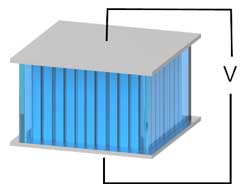 Researchers have created a heat-to-electricity device that runs on ions and which could someday harness the body's heat to provide energy.
Researchers have created a heat-to-electricity device that runs on ions and which could someday harness the body's heat to provide energy.
Mar 25th, 2019
Read more
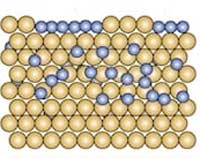 In the heat of a furnace, boron atoms happily dive into a bath of gold. And when things get cool, they resurface as coveted borophene.
In the heat of a furnace, boron atoms happily dive into a bath of gold. And when things get cool, they resurface as coveted borophene.
Mar 25th, 2019
Read more
 Researchers have developed a method based on artificial intelligence techniques that consider the atmospheric variations when designing the solar cells to produce more energy.
Researchers have developed a method based on artificial intelligence techniques that consider the atmospheric variations when designing the solar cells to produce more energy.
Mar 25th, 2019
Read more
 Researchers have found that certain organic semiconducting materials can transport spin faster than they conduct charge, a phenomenon which could eventually power faster, more energy-efficient computers.
Researchers have found that certain organic semiconducting materials can transport spin faster than they conduct charge, a phenomenon which could eventually power faster, more energy-efficient computers.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed