| Posted: Aug 29, 2017 |
Scientists move graphene closer to transistor applications
(Nanowerk News) Scientists at the U.S. Department of Energy’s Ames Laboratory were able to successfully manipulate the electronic structure of graphene, which may enable the fabrication of graphene transistors-- faster and more reliable than existing silicon-based transistors (Carbon, "Manipulation of Dirac cones in intercalated epitaxial graphene").
|
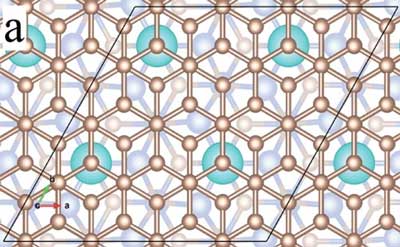 |
| The atomic and electronic structure of epitaxial graphene intercalated with Ytterbium. (Image: Ames Laboratory)
|
|
The researchers were able to theoretically calculate the mechanism by which graphene’s electronic band structure could be modified with metal atoms. The work will guide experimentally the use of the effect in layers of graphene with rare-earth metal ions “sandwiched” (or intercalated) between graphene and its silicon carbide substrate. Because the metal atoms are magnetic the additions can also modify the use of graphene for spintronics.
|
|
“We are discovering new and more useful versions of graphene,” said Ames Laboratory senior scientist Michael C. Tringides. “We found that the placement of the rare earth metals below graphene, and precisely where they are located, in the layers between graphene and its substrate, is critical to manipulating the bands and tune the band gap.”
|
|
Graphene, a two-dimensional layer of carbon, has been extensively studied by researchers everywhere since it was first produced in 2004 because electrons travel much faster along its surface, making it an ideal potential material for future electronic technologies. But the inability to control or tune graphene’s unique properties has been an obstacle to its application.
|
|
Density Functional Theory calculations predicted the configurations necessary to demonstrate control of the band gap structure. “Ames Laboratory is very good at synthesis of materials, and we use theory to precisely determine how to modify the metal atoms,” said Minsung Kim, a postdoctoral research associate. “Our calculations guided the placement so that we can manipulate these quantum properties to behave the way we want them to.”
|

