| Posted: Jun 30, 2017 | |
Mussel-inspired property of dopamin introduced to nanoparticles for tumor targetting(Nanowerk News) A group of researchers in Korea have synthesized an amphiphilic hyaluronic acid-ceramide-dopamine (HACE-d) conjugate and introduced the mussel-inspired properties of dopamine into the nanoparticles for tumor-specific delivery of phloretin and in vivo visualization of cancer. |
|
| The team reports their findings in ACS Applied Materials & Interfaces ("Mussel-Inspired Hyaluronic Acid Derivative Nanostructures for Improved Tumor Targeting and Penetration"). | |
 |
|
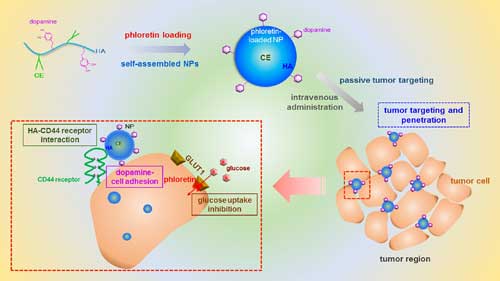
| Schemes regarding tumor targeting and penetration strategy of HACE-d/phloretin nanoparticles. Phloretin (as a hydrophobic drug) was loaded to self-assembled HACE-d nanoparticles. The physicochemical properties (i.e., particle size) of nanoparticles and HA-CD44 receptor interaction may provide passive and active tumor targetability after intravenous injection. In addition, dopamine-cell adhesion may enhance the tumor penetration efficiency of HACE-d nanoparticles. Phloretin may inhibit glucose uptake via GLUT1 thereby induce anticancer activities. (© ACS) (click on image to enlarge) | |
| Various tumor targeting strategies to deliver anticancer agents selectively to tumor region use an enhanced permeability and retention (EPR) effect to avoid rapid clearance of the nanocarriers from the bloodstream. | |
| Previously, researchers already have reported the mussel-inspired property of dopamine included in marine mussel adhesive proteins, and dopamine with catecholamine groups had been used for enhancing cellular adhesion. | |
| In this present work, the scientists fabricated HACE-d/phloretin nanoparticles with 279 nm mean diameter, 0.2 polydispersity index, and -18 mV zeta potential. Along with the HA-CD44 receptor binding-intermediated endocytosis of HACE-d nanoparticles, dopamine group exposed in the outer layer of nanocarriers seems to improve cellular accumulation efficiency and subsequent in vitro antitumor efficacy in MDA-MB-231 cells (a line of human breast cancer cells). | |
| Using a MDAMB-231 spheroid model, the researchers observed enhanced infiltration and superior spheroid growth inhibitory effect of HACE-d nanoparticles, in comparison with HACE nanoparticles. | |
| This means that, according to the results of real-time optical imaging studies in a tumor bearing mouse, improved tumor targeting and penetration can be achieved with HACE-d nanoparticles rather than HACE nanoparticles. | |
| The team points out that the HACE-d/phloretin nanoparticles group also exhibited improved anticancer activities, compared with their other experimental groups, in the tumor bearing mouse. | |
| "The combination of passive targeting (an enhanced permeability and retention effect), active targeting (HA-CD44 receptor interaction), improved cellular adhesion and tumor penetration of dopamine residue, and glycolysis inhibition of phloretin may be an unprecedented tumor targeting and penetration approach, and it can be successfully applied for the therapy and imaging of CD44 receptor and GLUT1-expressed cancers," the authors conclude. |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
