| Posted: Aug 28, 2017 | |
Direct water splitting with polymer nanosheets using visible light(Nanowerk News) Researchers show that 1,3-diyne-linked conjugated polymer frameworks are able to efficiently catalyze overall water splitting under visible light irradiation with apparent quantum efficiencies of as high as 10% at 420 nm. |
|
| In their paper in Advanced Materials ("Conjugated Microporous Polymer Nanosheets for Overall Water Splitting Using Visible Light"), a research team from China attributes the superior photocatalytic activity to the unique porous, sheet-like, highly conjugated structures with longrange charge delocalization, and sufficient surface reactions to prevent substantial charge recombination. | |
| Based on first-principles calculations, the team further elucidates the possible four-electron reaction pathways along with reactive sites on polymer photocatalysts for overall water-splitting reactions. | |
| Photocatalytic water splitting, which can directly convert sunlight to molecular hydrogen (H2) and oxygen (O2) from water, represents a promising route toward producing clean and renewable energy. | |
| However, many photocatalysts for overall water splitting still suffer from insufficient stability, low quantum efficiency, and limited tunability in optical and electronic properties. | |
| The Chinese team shows that 1,3-diyne-linked conjugated microporous polymer nanosheets (CMPNs) are unique semiconducting polymers for efficient visible-light-driven overall water splitting. They are synthesized from 1,3,5-tris-(4-ethynylphenyl)-benzene (TEPB) and 1,3,5-triethynylbenzene (TEB) via oxidative coupling reactions (in the below figure referred to as the PTEPB and PTEB for CMPNs synthesized using TEPB and TEB, respectively). | |
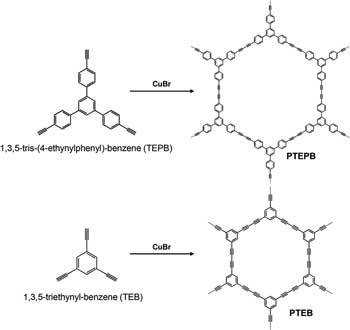 |
|
| Chemical structures of PTEPB and PTEB photocatalysts and the oxidative coupling reaction used to synthesize corresponding photocatalysts using CuBr as the catalyst at room temperature. (© Wiley-VCH Verlag) | |
| Furthermore, they demonstrate that the CMPNs are highly stable even after repeated visible photocatalytic tests, implying that the CMPNs can reduce charge build-up on the surface to prevent deleterious photochemical degradation. | |
| "Our findings provide the new insights in designing and synthesizing organic polymers capable of overall water splitting at neutral pH with sunlight as the only energy input," they conclude their report. |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
