| Posted: Nov 09, 2017 | |
Key to direct graphene transfer is the control of water intercalation(Nanowerk News) In new work, reported in ACS Applied Materials & Services ("Controlling Water Intercalation Is Key to a Direct Graphene Transfer"), researchers in Belgium demonstrate that interfacial water can insert between graphene and its growth substrate despite the hydrophobic behavior of graphene. |
|
| In their paper, the researchers discuss in detail graphene delamination from platinum (Pt) surfaces using electrochemical methods. They show that water intercalation between graphene and a Pt surface is critical to achieve a successful graphene delamination using an electrochemical method. | |
| They also demonstrate that intercalation effects can occur between graphene and a target wafer, resulting in unwanted graphene delamination effects. | |
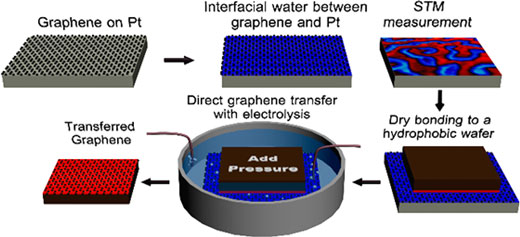 |
|
| The proposed process. (© ACS) | |
| Exposing a platinum/graphene sample for several days to ambient conditions or submerging it in warm ultrapure water gradually changes the graphene surface morphology and varies the chemical composition of the graphene/Pt interface. | |
| The results are consistent with water intercalation, which progressively develops at the interface. This intercalation process is essential to obtain a successful electrochemical graphene delamination procedure. | |
| Furthermore, the team established a direct graphene transfer process on the basis of interfacial water between graphene and the CVD growth substrate and of avoiding water intercalation between the hydrophobic target wafer and graphene. | |
| Such a direct graphene transfer avoids polymer contamination (no temporary support layer) and etching of the catalyst metal. As a result, recycling of the growth template becomes feasible. | |
| "Since the interaction between graphene and water is comparable to the interaction between MoS2 and water, a transfer procedure based on controlling water intercalation might also work for other 2D materials like MoS2," the authors conclude their report. "As a result, the proposed transfer process might even open the door for the predicted atomic-scale interlocking-toy-brick stacking of different hydrophobic 2D materials." |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
