| Posted: Jan 15, 2018 | |
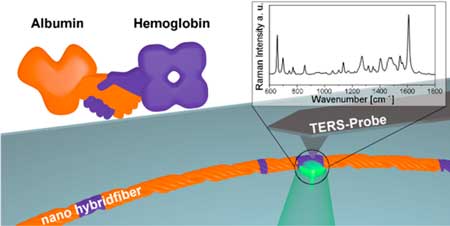
Protein handshake on the nanoscale(Nanowerk News) A new study published in ACS Nano ("Protein Handshake on the Nanoscale: How Albumin and Hemoglobin Self-Assemble into Nanohybrid Fibers") shows that it is possible to create hybrid protein nanofibers composed of two plasma proteins using a facile self-assembly approach supported by ethanol-induced protein denaturation. |
|
 |
|
| Creating and establishing proof of hybrid protein nanofibers, i.e., protein nanofibers that contain more than one protein, is a currently unsolved challenge in bioinspired materials science. Such hybrid protein nanofibers could serve as universal building blocks for the bottom-up preparation of functional materials with bespoke properties. | |
| In this work, the similarity in the amino acid sequences, in both human serum albumin (HSA) and hemoglobin (HGB) proteins, exposed upon conformational changes and their hydrophobic interactions are recognized as the driving force for the self-assembly mechanism of the hybrid protein nanofibers. | |
| The challenge of establishing the proof of hybrid protein nanofibers’ heterogenic nature can be overcome with nanospectroscopic techniques, such as TERS and AFM force spectroscopy. | |
| The German team of scientists believe that this self-assembly approach can successfully be applied to other plasma proteins assuming that the same amino acid sequences can be found in both proteins upon conformational changes. | |
| For example, the fibrinogen and fibronectin system is expected to form hybrid protein nanofibers due to the similarity in the primary structure and the existence of binding sites for fibrin in fibronectin. | |
| "Such hybrid protein nanofibers can be for example utilized as universal building blocks for the bottom-up preparation of larger bioactive structures or, on account of their high surface area, can be used as bioactive devices and sensors," the authors conclude. |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
