| Posted: January 23, 2007 |
A dynamic future for supramolecular chemistry |
|
(Nanowerk News) Jean-Marie Lehn of Louis Pasteur University, Strasbourg, France, winner of the Nobel Prize in Chemistry in 1987, shares his view of a dynamic future for supramolecular chemistry (From supramolecular chemistry towards constitutional dynamic chemistry and adaptive chemistry) - free access article.
|
|
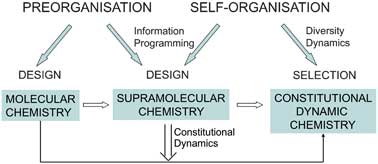
Beyond molecular chemistry, which is based on the covalent bond, supramolecular chemistry aims to develop highly complex chemical systems that interact through non-covalent intermolecular forces, such as hydrogen bonding or van der Waals interactions. During the past forty years supramolecular chemistry has grown into a major field, leading to the establishment of supramolecular science and technology.
|
|
Supramolecular chemists are now extending their research beyond the design of molecules that can be used for molecular recognition or catalysis. They are actively exploring systems that undergo self-organisation - systems that can spontaneously generate well-defined functional supramolecular architectures by self-assembly from their components. This spontaneous but controlled formation of nanoscale architectures could be used to engineer and process functional nanostructures, offering a powerful alternative to nanofabrication, going from construction to self-construction.
|
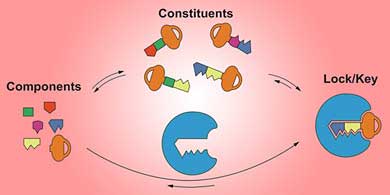 |
| Molecular fragments self-assemble to form a dynamic library of potentially bioactive compounds. (Image: RSC Publishing)
|
|
A further avenue of research for supramolecular chemists is constitutional dynamic chemistry. At a fundamental level, supramolecular chemistry is a dynamic chemistry. The interactions that connect the molecules of a supramolecular entity are easily broken and reformed, allowing supramolecular species to readily exchange their component parts. The same can be true for molecular chemistry - when a molecule contains covalent bonds that can form and break reversibly, its building blocks can be continuously exchanged and reorganised. These are the features that define constitutional dynamic chemistry on both the molecular and supramolecular levels.
|
 |
|
This dynamic form of chemistry can be put to use in areas as diverse as the development of dynamic materials and the search for biologically active compounds.
|
|
Dynamic materials can be defined as materials whose components are linked through reversible connections. They undergo spontaneous and continuous changes in makeup by assembly or disassembly processes. The intrinsic ability of dynamic materials to change their components means they can behave as adaptive or responsive materials.
|
|
For example, a molecular or supramolecular dynamic polymer, or dynamer, can incorporate and exchange different monomers, leading to changes in the polymer's properties. These polymers could be designed to select particular components in response to external stimulants such as heat or light.
|
|
The search for bioactive substances amounts to searching for a molecular key for a biological lock. Apart from the random screening of natural or synthetic compounds, there are two traditional approaches to this search: the rational design of a single correct key; or combinatorial chemistry, which relies on the generation of vast collections of keys that are tested by high-throughput screening.
|
|
Constitutional dynamic chemistry offers a third option - dynamic combinatorial chemistry. Dynamic libraries of potential keys can be generated from the reversible combinations of fragments of keys. The key that has either the strongest or the fastest-forming interaction with the lock is selected from all the possible combinations. This overall process, directed by the supramolecular lock and key interactions, bypasses the need to actually synthesise the constituents of a combinatorial library by letting the lock choose and assemble its optimal key.
|
|
The implementation of selection in chemistry is a fundamental change in outlook. Self-organisation by design strives to achieve full control over the target molecule or supramolecule, whereas self-organisation by selection takes advantage of dynamic diversity to allow variation in response to internal or external factors in a Darwinian fashion.
|
|
Constitutional dynamic chemistry paves the way towards an adaptive and evolutive chemistry, a further step towards unravelling the science of complex matter.
|


