| Nov 03, 2023 |
Researchers can now visualize osmotic pressure in living tissue
|
|
(Nanowerk News) In order to survive, organisms must control the pressure inside them, from the single-cell level to tissues and organs. Measuring these pressures in living cells and tissues in physiological conditions is a challenge.
|
|
In research that has its origin at UC Santa Barbara, scientists now at the Cluster of Excellence Physics of Life (PoL) at the Technical University in Dresden (TU Dresden), Germany, report in the journal Nature Communications ("In situ quantification of osmotic pressure within living embryonic tissues") a new technique to ‘visualize’ these pressures as organisms develop. These measurements can help understand how cells and tissues survive under pressure, and reveal how problems in regulating pressures lead to disease.
|
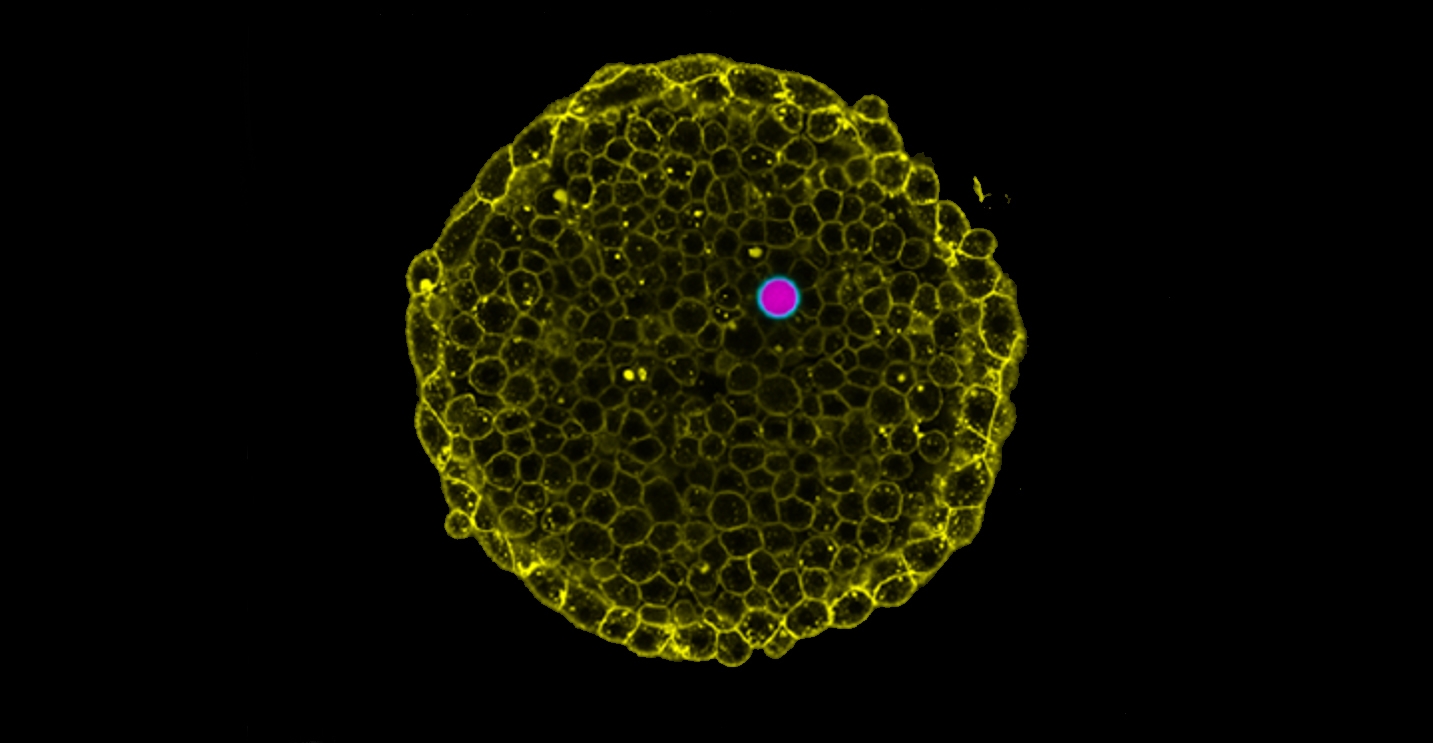 |
| Measurement of intracellular osmotic pressure. (Image: Courtesy of the researchers)
|
|
When molecules dissolved in water are separated into different compartments, water has the tendency to flow from one compartment to another to equilibrate their concentrations, a process known as osmosis. If some molecules cannot cross the membrane that separates them, a pressure imbalance — osmotic pressure — builds up between compartments. This principle is the basis for many technical applications, such as the desalination of seawater or the development of moisturizing creams. It turns out that maintaining a healthy functioning organism makes the list, too.
|
|
Our cells are constantly moving molecules in and out to prevent the pressure build-up from crushing them. To do so, they use molecular pumps that allow them to keep the pressure in check. This osmotic pressure affects many aspects of cells’ lives and even sets their size.
|
|
When cells team up to build our tissues and organs, they, too, face a pressure problem: Our vascular system, or organs such as the pancreas or liver, contain fluid-filled cavities known as lumens that are essential for their function. If cells fail to control osmotic pressure, these lumens may collapse or explode, with potentially catastrophic consequences for the organ. To understand how cells regulate pressure in these tissues, or how they fail to do so in disease, it is essential to measure and ‘see’ the osmotic pressure in live tissues. But unfortunately, this was not possible.
|
|
Until now.
|
|
Led by former UCSB professor Otger Campàs, who now holds the Chair of Tissue Dynamics at TU Dresden and is currently the managing director of PoL, the scientists devised a novel technique to measure the osmotic pressure in living cells and tissues by using special droplets known as double emulsions. For this pressure sensor, they introduced a water droplet into an oil droplet that permits water to flow through. When these “double-droplets” were exposed to salt solutions of different concentrations, water flowed in and out of the internal water droplet, changing its volume, until pressures were equilibrated. The researchers showed that the osmotic pressure can be measured by simply checking the droplet size. They then introduced these double-droplets into living cells and tissues using glass microcapillaries to reveal their osmotic pressure.
|
|
“It turns out that cells in animal tissues have the same osmotic pressure as plant cells but, unlike plants, they must balance it constantly with their environment to avoid exploding, since they do not have rigid cell walls,” Campàs said.
|
|
With this simple concept, this ingenious method now allows scientists to “see” osmotic pressure in a wide range of settings. “We know that several physical processes affect the working of our bodies,” Campàs said. “ In particular, osmotic pressure is known to play a fundamental role in the building of organs during embryogenesis, and also in the maintenance of healthy adult organs. With this new technique, we now can study how osmotic pressure impacts all these processes directly in living tissues.”
|
|
Beyond offering insights into the biological processes and physical principles that govern life, this method holds promising industrial and medical applications, including monitoring skin hydration, characterization of creams or foods, and diagnosis of diseases known to have osmotic pressure imbalances, such as cardiovascular diseases or tumors. The patent for this technique is currently being issued by UC Santa Barbara, where Campàs performed his research before joining TU Dresden.
|
|
Campàs’s lab previously developed unique techniques to measure the tiny forces that cells create inside tissues and also additional physical properties using minuscule single droplets. Antoine Vian, the lead author of the work and an expert in microfluidics, the technology that enables the generation of double-emulsion droplets, emphasized their key role.
|
|
“Double-emulsions are very versatile, with many different applications in science and technology,” he said. “Single droplets can be deformed, but are incompressible and do not allow pressure measurements. In contrast, double emulsion droplets can change size and be used as osmotic pressure sensors. Their use in living systems will surely enable new and exciting discoveries.”
|

