| Feb 20, 2024 |
It's the spin that makes the difference
|
|
(Nanowerk News) Biomolecules such as amino acids and sugars occur in two mirror-image forms – in all living organisms, however, only one is ever found. Why this is the case is still unclear. Researchers at Empa and Forschungszentrum Jülich in Germany have now found evidence that the interplay between electric and magnetic fields could be at the origin of this phenomenon.
|
 |
| Biomolecules such as our genetic material, DNA, basically exist in two mirror-image forms; however, all living organisms only ever use one of them. Why this is the case is still unclear. (Image: Adobe Stock)
|
|
The so-called homochirality of life – the fact that all biomolecules in living organisms only ever occur in one of two mirror-image forms – has puzzled a number of scientific luminaries, from the discoverer of molecular chirality, Louis Pasteur, to William Thomson (Lord Kelvin) and Nobel Prize winner Pierre Curie.
|
|
A conclusive explanation is still lacking, as both forms have, for instance, the same chemical stability and do not differ from each other in their physico-chemical properties. The hypothesis, however, that the interplay between electric and magnetic fields could explain the preference for one or the other mirror-image form of a molecule – so-called enantiomers – emerged early on.
|
|
It was only a few years ago, though, that the first indirect evidence emerged that the various combinations of these force fields can indeed "distinguish" between the two mirror images of a molecule. This was achieved by studying the interaction of chiral molecules with metallic surfaces that exhibit a strong electric field over short distances.
|
|
The surfaces of magnetic metals such as iron, cobalt or nickel thus allow electric and magnetic fields to be combined in various ways – the direction of magnetization is simply reversed, from "North up – South down" to "South up – North down".
|
|
If the interplay between magnetism and electric fields actually triggers "enantioselective" effects, then the strength of the interaction between chiral molecules and magnetic surfaces should also differ, for example – depending on whether a right-handed or left-handed molecule "settles" on the surface.
|
Mirror images prefer opposing magnetic fields
|
|
And this is indeed the case, as a team of researchers led by Karl-Heinz Ernst from the Empa's Surface Science and Coating Technologies lab and colleagues at the Peter Grünberg Institute at the Forschungszentrum Jülich in Germany recently reported in the scientific journal Advanced Materials ("Enantioselective adsorption on magnetic surfaces").
|
 |
| If only left-handed helicene spirals are deposited on the cobalt-copper surface, they clearly prefer cobalt islands with a certain direction of magnetization. In the image, the two triangular cobalt islands have opposite magnetization; the left-handed helicene molecules bind almost exclusively to the island on the right and avoid the island on the left (except for a few molecules at the edge of the island). (Image: Peter Grünberg Institute/Jülich)
|
|
The team coated a (non-magnetic) copper surface with small, ultra-thin "islands" of magnetic cobalt and determined the direction of the magnetic field in these using spin-polarized scanning tunneling microscopy; as mentioned before, this can run in two different directions perpendicular to the metal surface: North up or South up. They then deposited spiral-shaped chiral molecules – a 1:1 mixture of left- and right-handed heptahelicene molecules – onto these cobalt islands in ultrahigh vacuum.
|
|
Then they "simply" counted the number of right- and left-handed helicene molecules on the differently magnetized cobalt islands, almost 800 molecules in total, again using scanning tunneling microscopy. And lo and behold: Depending on the direction of magnetic field, one or the other form of the helicene spirals had settled preferentially (see right side of the graphic).
|
|
Moreover, the experiments showed that the selection – the preference for one or the other enantiomer – not only occurs during the binding on the cobalt islands, but already beforehand. Before the molecules take up their final (preferred) position on one of the cobalt islands, they migrate long distances across the copper surface in a significantly weaker bound precursor state in "search" for an ideal position. They are only bound to the surface by so-called van der Waals forces. These are merely caused by fluctuations in the electronic shell of atoms and molecules and are therefore relatively weak. The fact that even these are influenced by magnetism, i.e. the direction of rotation (spin) of the electrons, was not known thus far.
|
Electrons with the "wrong" spin are filtered out
|
|
Using scanning tunneling microscopy, the researchers were also able to solve another mystery, as they reported in the journal Small last November. Electron transport – i.e. electric current – also depends on the combination of molecular handedness and magnetization of the surface. Depending on the handedness of the bound molecule, electrons with one direction of spin preferentially flow – or "tunnel" – through the molecule, meaning that electrons with the "wrong" spin are filtered out.
|
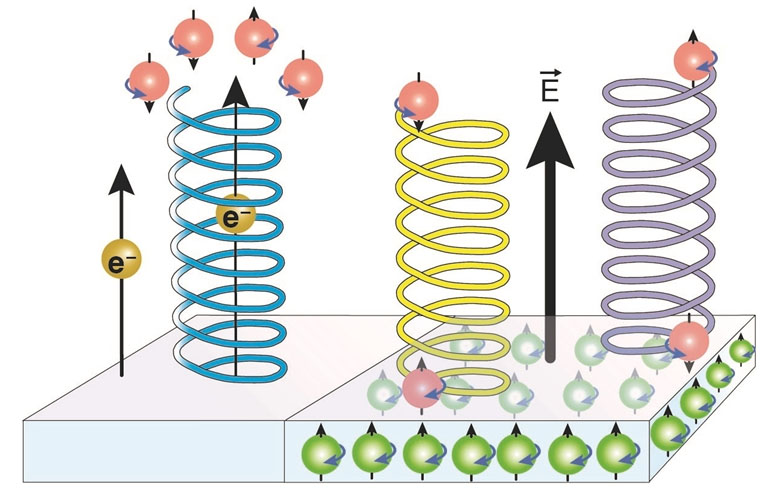 |
| This is how chirality-induced spin selectivity (CISS effect) manifests itself: Electrons (e– or red and green spheres with arrows indicating electron spin, either up or down) with the "wrong" direction of rotation (spin) are held back or filtered out when tunneling through spiral molecules, depending on the handedness of the spirals (left- or right-handed), so that one type of electron spin predominates (electrons with the arrow pointing downwards on the left side). The electric field of a metallic surface (E, pointing upwards, right side) shifts the electrons in the bound heptahelical molecules; these accumulate slightly in the lower part of the molecule near the surface. In the case of chiral molecules, electrons with different spins are also shifted differently depending on the handedness of the molecule. The molecule becomes "spin-polarized", i.e. also magnetic. Depending on the direction of magnetization of the metallic surface, chiral molecules therefore interact with it to different degrees. In this example, the purple spiral therefore binds more strongly to the surface than the yellow one, as opposite spins "attract" each other (the red and green electrons with different spins sitting on top of each other). (Graphic: Empa)
|
|
This chirality-induced spin selectivity (CISS effect, see left side of the graphic) had already been observed in earlier studies, but it remained unclear whether an ensemble of molecules is necessary for this or whether individual molecules also exhibit this effect. Ernst and his colleagues have now been able to show that individual helicene molecules also exhibit the CISS effect. "But the physics behind this is still not understood," admits Ernst.
|
|
The Empa researcher also believes that his findings eventually cannot fully answer the question of the chirality of life. In other words, the question that the Nobel Prize winner in chemistry and ETH chemist Vladimir Prelog described as "one of the first problems of molecular theology" in his Nobel Prize lecture in 1975. But Ernst can imagine that in certain surface-catalyzed chemical reactions – such as those that could have taken place in the chemical "primordial soup" on the early Earth – a certain combination of electric and magnetic fields could have led to a steady accumulation of one form or another of the various biomolecules – and thus ultimately to the handedness of life.
|



