| Nov 01, 2017 |
One-step 3D-printing of catalysts
|
|
(Nanowerk News) Researchers at Ames Laboratory have developed a 3D printing process that creates a chemically active catalytic object in a single step, opening the door to more efficient ways to produce catalysts for complex chemical reactions in a wide scope of industries (ACS Catalysis, "Direct 3D Printing of Catalytically Active Structures").
|
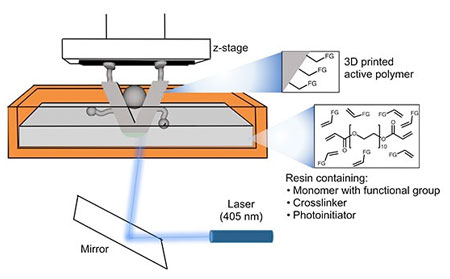 |
| Catalysts can be built in one step by directly shining a laser through a bath of customized resins that polymerize and harden layer-by-layer. (Image: Ames Laboratory)
|
|
While 3D printing has found applications in many areas, its use as a way to control chemical reactions, or catalysis, is relatively new. Current production of 3D catalysts typically involves various methods of depositing the chemically active agents onto pre-printed structures.
|
|
The Ames Laboratory method combines the structure with the chemistry in only one step using inexpensive commercial 3D printers. The structures are designed in a computer and built directly by shining a laser through a bath of customized resins that polymerize and harden layer-by-layer. The final product that emerges has catalytic properties already intrinsic to the object.
|
|
“The monomers, or building blocks that we start with, are designed to be bifunctional. They react with light to harden into the three-dimensional structure, and still retain active sites for chemical reactions to occur,” said Sebastián Manzano, a graduate student in the Department of Chemistry at Iowa State and who conducted most of the experiments.
|
 |
| Ames Laboratory has developed a one-step 3D-printing process for catalysts that can be customized to any shape-- in this demonstration the Ames Laboratory logo design. (Image: Ames Laboratory)
|
|
The catalysts built with this method demonstrated success in several reactions common to organic chemistry. They are also adaptable with further post-processing, making possible multi-step reactions.
|
|
“We can control the shape of the structure itself, what we call the macroscale features; and the design of the catalyst, the nanoscale features, at the same time”, said Igor Slowing, a scientist in heterogeneous catalysis at the U.S. Department of Energy’s Ames Laboratory. “This opens up many possibilities to rapidly produce structures custom designed to perform a variety of chemical conversions.”
|


