| Posted: Jun 23, 2009 | |
Humidity doesn't interfere with this nanotechnology gas sensor |
|
| (Nanowerk Spotlight) Gas sensing applications are numerous in our modern society and include process monitoring, environmental compliance, health applications, homeland security, agriculture, etc. Gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Thus, the more surface available, the more sensitive the sensor will be. Nanoscale materials – particles, nanotubes and nanowires – are intriguing materials for next-generation nanotechnology gas sensors since their relative surface areas are so large (for more on this topic read: "Bomb sniffing dogs today, nanotechnology tomorrow" and "Nanotechnology puts your nose on a carbon nanotube "). | |
| A problem with existing gas nanosensors is the cross-interference of other gas analytes. For instance, carbon nanotube based gas sensors for the prominent air pollutant nitrogen dioxide have shown strong interference of ethanol and ammonia gases to the NO2 response. Another cross-interference often is caused by humidity, i.e. the water vapor in the air. | |
| New research now demonstrates how the manufacturing of a nanosensor for ammonia gas can be tuned to eliminate the interference of water vapor. The trick lies in accurately controlling the synthesis of the sensing nanomaterial. | |
| "By precisely functionalizing single-walled carbon nanotube (SWCNT) networks with camphorsulfonic-acid-doped polyaniline (CSA-doped PANI), the opposite electrical responses toward humid air of CSA-doped PANI and SWCNTs effectively canceled the humidity interference," Marc Deshusses explains to Nanowerk. "Our results demonstrate that appropriate selection of nanomaterials and fine tuning of the synthesis conditions can overcome some of the limitations encountered by nanosensors." | |
| Deshusses is a professor in the Department of Civil and Environmental Engineering at Duke University. Together with Nosang Myung and colleagues from his former position at the Department of Chemical and Environmental Engineering at UC Riverside, and UC Riverside's Center for Nanoscale Science and Engineering, he has published his findings in the June 2, 2009 online edition of Nanotechnology ("A gas nanosensor unaffected by humidity"). | |
 |
|
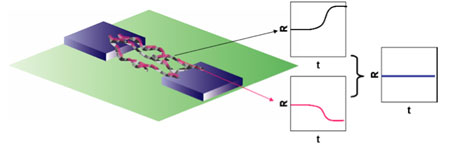
| The fabrication of a humidity independent gas nano-sensor for ammonia is demonstrated. By functionalizing single-walled carbon nanotube (SWCNT) networks with a precise amount of camphor-sulfonic acid-doped polyaniline (PANI(CSA)), the opposite electrical resistance responses of CSA-doped PANI and SWNTs toward humid air effectively canceled each other, thereby eliminating any effects of humidity during sensing experiments. (Image: Dr. Deshusses, Duke University) | |
| The team exploited the fact that bare SWCNTs have a positive response to relative humidity while the PANI functionalized SWCNTs have a negative response to relative humidity. Striking the right balance in synthesizing the sensing material, where it would show a zero response to relative humidity, was a question of precisely controlling the thickness of the electropolymerized CSA-doped PANI onto the nanotubes. | |
| Deshusses notes that their sensors showed excellent sensitivity toward ammonia at room temperature with minimum interference from water vapor. "Overall, our results demonstrate that shortcomings of conventional sensors can be overcome by designing novel nanoengineered materials" he says. "The approach of using nanostructures with opposite electrical responses to interferences should be generally applicable to the development of analyte specific nanosensors. Our approach paves the way for the development of more selective gas nanosensors." | |
 |
|
| AFM images and diameter histograms of CSA-doped PANI coated SWCNT networks as a function of deposition time: (A) bare carboxylated SWNT network, (B) after 2 min PANI deposition, and (C) after 5 min deposition. Each histogram was built from 100 individual SWCNTs transects. (Reprinted with permission from IOP Publishing) | |
| In their effort to fabricate a reliable low-cost sensor, the team selected electropolymerization to decorate the SWCNTs. This is a simple and cost-effective technique which offers spatially-tailored functionalization. | |
| To conduct gas detection studies, the researchers wire bonded the sensors and connected them in series with a load resistor. They then experimented with various sensors that were fabricated with different electrodeposition times until they found the one whose resistance was virtually independent of a given amount of relative humidity. | |
| After obtaining a steady state response, the sensors were purged with humid air for recovery. Deshusses says that the response to ammonia was reversible although recovery was slow compared to other nanomaterials-based sensors (in general, response times ranging from 10 to 30 minutes have been reported by others for organic polymer functionalized CNT nanosensors). | |
| The next challenge for Deshusses and his team lies in making faster, more sensitive, and more stable sensors. For these sensors to be used commercially one day, they also need to find a way to mass produce these sensors in a reliable fashion. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
