| Posted: Aug 11, 2009 | |
Bio-interface nanosensor visually reports damage to a material |
|
| (Nanowerk Spotlight) The term bio-interface describes the boundary between synthetic materials such as plastics, and biological systems. This rapidly growing research area, where biology and material sciences overlap, is creating new opportunities for the design, synthesis, and optimization of biologically-enabled and biologically-inspired materials. It involves manufacturing and characterization of functional surfaces for specific interactions with bio-systems and studies of the molecular and kinetic processes occurring at such interfaces, ranging from small molecule and biomolecular interactions, to cell adhesion, differentiation and tissue formation at the interface. | |
| For example, the incorporation of proteins into polymers can result in hybrid materials that combine the properties of the polymer as a cost-effective and easy to process material with the highly evolved biological functionality of the protein, enabling new concepts for construction of sensors and biomedical materials. | |
| While researchers so far have been focusing on altering the properties of a polymer by adding the functionality of a biomolecule, a group in California has now demonstrated the reverse situation, where changes in the polymer can alter the properties of the protein. They report a polymer–protein hybrid material in which changes in stress of the polymer matrix result in changes to the fluorescence properties of the protein complex, thus creating a material that visually reports damage. | |
| Douglas S. Clark, a professor in the Department of Chemical Engineering at the University of California Berkeley and his lab have recently proven the concept of a protein-based nanosensor that is able to report deformation of the embedding polymer matrix. The team combined the structural properties of a chaperonin from a thermophilic organism with the spectral properties of fluorescent proteins in order to generate a protein complex that exhibits fluorescent energy resonance transfer (FRET) and is sensitive to structural deformation. Such self-reporting materials could be used in myriad applications where easy and early detection of damage is essential to avoid catastrophic failure of the material. | |
| Clark and his team have reported their findings in a recent paper in Angewandte Chemie International Edition ("'Mechanical Nanosensor Based on FRET within a Thermosome: Damage-Reporting Polymeric Materials"). | |
| In their work, the scientists combined the structural properties of the thermosome (THS), a chaperonin (protein complex) from the thermophilic organism Thermoplasma acidophilum, with the spectral properties of fluorescent proteins to generate a protein complex that exhibits fluorescence resonance energy transfer (FRET) and is sensitive to structural deformation. | |
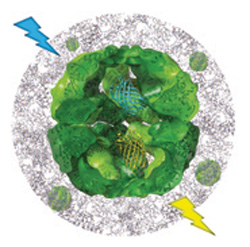
|
|
| Mechanical nanosensor based on FRET within a thermosome for damage-reporting polymeric materials. (Image: Clark Lab, UC Berkeley) | |
| "Our concept for a THS-based stress sensor is based on the idea that guest proteins can be permanently entrapped into the cavities by covalent linker chemistry," writes Clark. "Entrapping a donor–acceptor pair of fluorescent proteins into the cavities gives the possibility for FRET to occur. The centers of the two cavities are approximately 7 nm apart, which is close to the Förster radius (4.9 nm) of enhanced cyan fluorescent protein (eCFP) and enhanced yellow fluorescent protein (eYFP), which are the most commonly used donor–acceptor FRET pair in molecular biology. We thus hypothesized that, by covalently crosslinking chains of a polymer matrix with the THS–guest complex, a sufficient force applied to the matrix will separate the two halves of the THS, which results in reduced FRET." | |
| The coupling between the modified THS and the modified fluorescent proteins was carried out by simple incubation of a mixture of the proteins for several hours at room temperature in a coupling buffer. Subsequent fluorescence spectroscopy measurements proved that FRET occurs between the fluorescent proteins. | |
| The researchers then went on to investigate the effect of mechanical deformation of the polymer on the biomechanical sensor. To this end, they strained samples of the polymer until they fractured and then imaged the area surrounding the fracture by multichannel confocal microscopy. | |
| Clark's team found that the FRET efficiency in the non deformed material surrounding the cracks is surprisingly low. "However, the FRET signal increases 1.5-fold in regions corresponding to the locations of the cracks. The FLIM images show a shorter mean lifetime of the donor within the cracks compared to the surrounding matrix , which corresponds to a higher FRET efficiency. The lifetime difference suggests that changes in the microenvironment of the fluorophores within the cracks occurs, which most likely arises from a shortening of the donor–acceptor distance." | |
| Further work is now underway in Clark's lab to examine the sensitivity of the sensor. Ideally, it will be able to report damage on a submicrometer scale before visible cracks become detectable by standard microscopic methods. These self-reporting materials could be used in myriad applications where easy and early detection of damage is essential to avoid catastrophic failure of the material. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
