| Posted: Mar 02, 2007 | |
The challenge of designing nanomaterials with reduced toxicity |
|
| (Nanowerk Spotlight) Obtaining an understanding, at the atomic level, of the interaction of nanomaterials with biological systems has recently become an issue of great research interest. Nanomaterials can exhibit drastically different characteristics compared to their bulk counterparts. | |
| Although the use of such materials in biological systems opens avenues for the creation of novel biosensing and alternative nanomedical technologies, these nanomaterials can also be highly toxic. A greater understanding of the interaction of nanomaterials with biological systems, especially of the interaction of nanomaterials with cell membranes, will enable scientists to take full advantage of the unique properties of nanomaterials while minimizing their adverse effects. | |
| Fullerenes and their derivatives are an important subset of nanomaterials. Fullerenes have been used as robust oxygen scavengers, anti-HIV drugs, X-ray contrast agents, and transporters for delivering antibodies. While experimental studies suggest that the toxicity of nanomaterials depends critically on their surface properties, it was also found that, in the case of fullerenes, functionalizing the molecules can reduce their toxicity notably. | |
| New work by U.S. researchers offers a mechanistic view on the different cytotoxicity of fullerenes and their functionalized derivatives - a first in this important field of nanotoxicity. The major finding is that pristine fullerene can readily jump into a lipid bilayer while the translocation of a functionalized fullerene is severely hindered due to its surface charge, leading to a much reduced toxicity. | |
| It has been known for a while that surface functionalization could modify the toxicity of nanomaterials. However, the mechanisms behind these observations are not well understood. This situation has motivated researchers to find a mechanistic explanation by taking a common nanomaterial – a fullerene – and study its interaction with a common biological entity – a cell. | |
| Existing studies on the interactions of C60 with cell membranes are limited, but it has been suggested that the accumulation of C60 in a DPPC bilayer may cause degradation and a suppression of the phase transitions of the host bilayer. Back in 2004, Colvin’s group at Rice University determined that the toxicity of pristine C60 can cause membrane leakage ("The Differential Cytotoxicity of Water-Soluble Fullerenes"). They also found that the functionalization of C60 dramatically lowered toxicity with no resultant membrane leakage. | |
| "Studies of these atomistic processes have only received recent attention and the effects of surface functionalization have yet to be addressed" Dr. Rui Qiao tells Nanowerk. " Now we have conducted what could be the first comparative atomistic study of effects of surface functionalization on toxicity of nanomaterials. Our simulations indicate that surface functionalization can significantly modify the interactions between fullerenes and cell membranes. The results is a very different translocation behavior and impact on membrane structure." | |
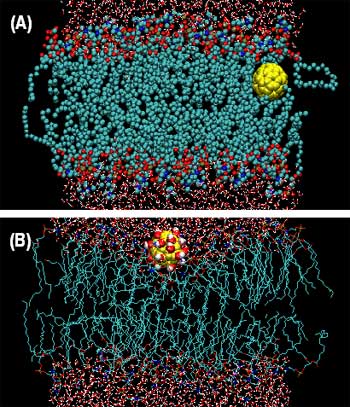 |
|
| A): pristine fullerene adsorbed inside the membrane bilayer, (B) functionalized fullerene adsorbed on the membrane bilayer (Image: Dr. Qiao, Clemson University) | |
| Qiao, an assistant professor in the Department of Mechanical Engineering at Clemson University, is also first author of a recent paper in Nano Letters, published online on February 23, 2007 ("Translocation of C60 and Its Derivatives Across a Lipid Bilayer"). | |
| This paper details the study by the Clemson researchers of the interaction of pristine and functionalized C60 with a lipid bilayer and the subsequent translocation of these fullerenes using molecular dynamics simulations. These simulations were originally conducted to match Dr. Pu-Chun Ke's experimental study on fullerene translocation (not published yet; we'll cover it in an upcoming Spotlight) and Dr. Stephen Klaine's nanotoxicity study ("In vivo Biomodification of Lipid-Coated Carbon Nanotubes by Daphnia magna"). Both Ke and Klaine are co-authors of the paper. | |
| The interesting results of this new study are twofold, Qiao tells Nanowerk: | |
| "1) While a pristine fullerene can jump into a cell's membrane bilayer at a mean speed of several meters per second and translocate the bilayer quickly, its functionalized counterpart can hardly enter the membrane and the translocation time is orders of magnitude longer. | |
| 2) While a pristine fullerene adsorbed in the bilayer can facilitate the formation of micro-pores on the membrane, the effects of its functionalized counterpart are the opposite." | |
| These observations offer a mechanistic explanation on the reduced toxicity of functionalized fullerenes. Furthermore, the findings reported in this paper show that numerical simulations can be a useful tool in studying nanotoxicity and complement experimental investigations. Ultimately, these insights could facilitate the design of nanomaterials with reduced toxicity. | |
| The Clemson researchers point out that the greatest challenge in terms of numerical modeling is the lack of efficient simulation techniques that are capable of capturing the large scale of biological responses to nanomaterials. | |
| "The interactions between nanomaterials and biological systems occur from the nanoscale up and are far more complex than the biophysical aspect we have addressed in this research" says Qiao. "Modeling such processes with full details is beyond the capability of current simulation techniques." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
