| Posted: Apr 06, 2010 | |
Controlling the properties of boron nitride nanoribbons with hydrogenation |
|
| (Nanowerk Spotlight) Boron nitride (BN) is a synthetic white solid material which remained undeveloped as a commercial material until the latter half of the 20th century. Boron and nitrogen are neighbors of carbon in the periodic table (in combination boron and nitrogen have the same number of outer shell electrons) and the atomic radii of boron and nitrogen are similar to that of carbon. It is not surprising, therefore, that boron nitride and carbon exhibit similarity in their crystal structure. | |
| Consequently, researchers have been exploring BN nanomaterials – from nanotubes to nanosheets and nanoribbons – and found similarities, but also differences to the the properties of the corresponding carbon nanomaterials. BN nanotubes, for instance, have many of the excellent properties of the well-known carbon nanotubes (CNTs) because they share the same structure. BN nanotubes were found to have high chemical stability, high resistance to oxidation at high temperatures and are a stable wide-band-gap semiconductor which makes them useful for applications at high temperatures or in corrosive environments (see "Nanotechnology in space"). | |
| Similarly, motivated by the outstanding properties of graphene, the boron nitride single layer – a structural analogy of graphene – has been extensively studied both theoretically and experimentally, and has been experimentally realized. | |
| Cutting the boron nitride single layer makes it possible to obtain BN nanoribbons (BNNRs). These BN-based nanomaterials also exhibit high thermal and chemical stability, and have raised great expectations for applications for instance in nanoscale electronic devices. | |
|
"However, BN nanomaterials, from nanotubes to nanosheets and nanoribbons, are wide-band-gap semiconductors, and their band structures are rather robust, and difficult to modulate, which is a substantial obstacle for their applications in nanoelectronic devices," | |
| Only recently, researchers have demonstrated that graphene's properties can be controlled by reversible hydrogenation – resulting in graphane (see: "Researchers convert graphene into its hydrogenated equivalent graphane"). This work has been the first to show that a chemical approach can be used to tailor the properties of a nano-material like graphene, in order to tune it to a particular application. | |
| In a recent paper in Journal of the American Chemical Society ("Hydrogenation: A Simple Approach To Realize Semiconductor-Half-Metal-Metal Transition in Boron Nitride Nanoribbons"), an international team led by Chen and Zhen Zhou, an associate professor at Nankai University in PR China, predicted that hydrogenation might also be a simple approach to tune the band structure of graphene-like boron nitride structures. | |
 |
|
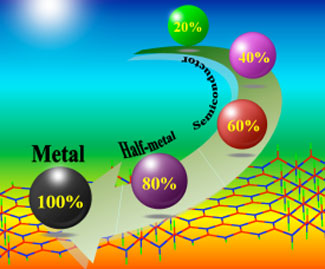
| The electronic properties of the partially hydrogenated zigzag BNNRs with different hydrogenation percentages. (Image: Dr. Wei Chen, University of Puerto Rico) | |
| Using large-scale first principles computations, the team was able to reveal that the fully hydrogenated zigzag BNNRs are ferromagnetic and metallic, and the partially hydrogenated zigzag BNNRs – using hydrogenated BNNRs and pristine BNNRs as building units – exhibit diverse electronic and magnetic properties: they are nonmagnetic semiconductors when the percentage of hydrogenated BNNR blocks is minor, while a semiconductor→half-metal→metal transition occurs, accompanied by a nonmagnetic→magnetic transfer, when the hydrogenated part is dominant. | |
| "This means" explains Chen, "that controlling the hydrogenation ratio can precisely modulate the electronic and magnetic properties of zigzag BNNRs, which endows BN nanomaterials many potential applications in the area of novel integrated functional nanodevices." | |
| These findings by Chen and Zhou may guide researchers to fabricate diverse electronic and magnetic properties in unitary BN-based nanomaterials in future experiments. | |
| "Based on the novel tunable electrical properties of boron nitride, we can envision many potential applications including biosensors, supercapacitor, fuel cells, etc., using these BN materials," comments Chen's co-author Chenzhong Li from the Nanobioengineering/Bioelectronics Lab at Florida International University. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
