| Posted: Jul 28, 2010 | |
'Smart' sand: grain-sized nanotechnology electronic noses are on the horizon |
|
| (Nanowerk Spotlight) Imagine a device the size of – and nearly as cheap as – a grain of sand which is capable of analyzing the environment around it, recognize its chemical composition, and report it to a monitoring system. This is the concept of nanotechnology-based electronic noses (e-nose) – miniature electronic devices which mimic the olfactory systems of mammals and insects and which will lead to better, cheaper and smaller sensor devices (read more: "Nanotechnology electronic noses"). | |
| An international team of researchers has made a further step towards this vision and demonstrated a novel analytical sensor which mimics our olfaction system. The difference between this and similar prior e-noses is that the active element of this new device is an individual wedge-like nanowire (nanobelt) made of tin dioxide. The required diversity of the sensing elements is encoded in the nanobelt morphology via longitudinal width variations of the nanobelt realized during its growth and via functionalization of some of the segments with palladium catalyst. | |
| "Our approach demonstrates the potential of combining bottom-up nanowire fabrication protocols with state-of-the art microfabrication methods to design prospective simple sensing arrays which, in principle, might be scaled down to the size of few micrometers and thus become the smallest analytical instrument," Andrei Kolmakov, an associate professor in the physics department at Southern Illinois University at Carbondale, tells Nanowerk. | |
| Kolmakov and a team of researchers from Karlsruhe Institute of Technology, Rensselaer Polytechnic Institute, Sincrotrone Trieste, and first author Victor V. Sysoev from Saratov State Technical University, have published their findings in the July 20, 2010 online issue of ACS Nano ("Single-Nanobelt Electronic Nose: Engineering and Tests of the Simplest Analytical Element"). | |
| In what probably is the simplest and yet fully functioning e-nose, the device is made of an individual single-crystal metal oxide quasi-1D nanobelt. The nanobelt was indexed with a number of platinum electrodes in a way that each segment of the nanobelt between two electrodes defines an individual sensing elemental receptor of the array. | |
 |
|
| One of the possible device architectures (not actual device used): morphologically encoded nanostructure in contact with array of metal electrodes. Each segment has a different resistance what will provide the required diversity in sensing and as a result the recognition capability. (Image: Dr. Kolmakov, Southern Illinois University at Carbondale) | |
| "We contacted this individual nanobelt with dozen of electrodes and obtained an array of sensors" explains Kolmakov. "Due to this variation of the resistance along the length on the nanobelt the same gas will produce slightly different responses, which leads to the nanobelt's recognition ability. In fact, this is similar to our own human olfactory receptors which are not very gas selective individually but slightly differ from each other." | |
| Previous designs of nanowire-based electronic noses were using different nanowires to obtain different responses. That meant that they were more complex, expensive, fragile etc. Compared to this, nothing can be simpler, cheaper, and more robust than just a single individual nanowire. | |
| "Our fabrication advance can be compared with development of integrated electronics versus circuits based on individual electronic components" says Kolmakov. "In our case the main sensor functionality is integrated into one single nanostructure." | |
| The research team based their device on tin oxide. This is a well studied semiconducting oxide which traditionally serves as a testbench material to implement novel sensing principles and platforms in gas sensorics. Using a conventional vapor-solid method they grew tin oxide nanobelts and selectively doped segments of the nanobelt's surface with palladium nanoparticles in order to enhance the resistivity of these segments. | |
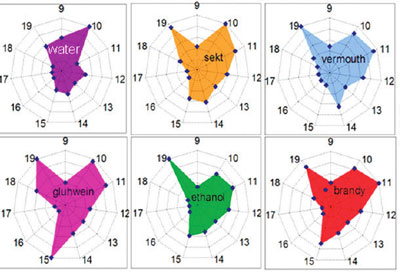
| The real-world applications of e-nose instruments would have to deal with complex odors or aromas, for example, ones widespread in the food industry. To test the performance of their e-nose against such complex environments, the researchers exposed a 10-segment single nanobelt sensor to the vapors from the headspace of four alcoholic beverages (glühwein, sparkling wine, vermouth, brandy). To eliminate the strong influence of the different ethanol content to the sensor signal, all beverages were diluted in distilled water to contain the same amount (10 rel. %) of ethanol. The resistance of all 10 segments of the tested nanobelt were plotted in a radar diagram and the results showed that the results are clearly analyte specific. | |
 |
|
| Polar plots of the responses of the nanobelt multisensor array to the aroma of various alcohol beverages. (Reprinted with permission from American Chemical Society) | |
| Kolmakov notes that the potential applications for this sensor device are are numerous and include stand alone truly microscopic detectors and analyzers which can be part of RFID tags (radio-frequency identification devices), smart phones, military devices (micro vehicles), medicine etc. | |
| The researchers' vision is to be able to grow 'smart' sand – imbed a battery, simple circuitry and reporter all on to the same nanostructure. | |
| "What is amazing is that separately all these tasks already have been realized" says Kolmakov. "We just need to combine them into one nanodevice." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
