| Posted: Sep 27, 2010 | |
Single nanowires provide unique tool for nanoscale battery diagnosis |
|
| (Nanowerk Spotlight) Traditionally, battery materials have usually been studied with bulk quantities in a complex environment with both active electrode components and many other supporting materials such as polymer binders and conductive additives. Although nanomaterials have been found to be able to improve battery performance, the complexity has made it hard to tell clearly about their advantages. Moreover, it is difficult to know whether fast capacity fading is due to the intrinsic nature of the transport property changes of active nanomaterials or an extrinsic reason from their interactions with the supporting materials, if all of them are studied together. | |
| The goal to understand the intrinsic reason of active material capacity fading has motivated a group of researchers to design single nanowire electrochemical devices as an extremely simplified model system to push the fundamental limits of the nanowire materials for energy storage applications. The result is a powerful and effective diagnostic tool for property degradation of lithium ion based energy storage devices. | |
| In the September 10, 2010 online edition of Nano Letters ("Single Nanowire Electrochemical Devices"), they report a study of vanadium oxide based cathode and silicon based anode at the single nanowire level and demonstrated that a single nanowire electrode can work as a versatile platform to study the correlation between material structure changes, transport property, and electrochemical property. | |
| "In our work, the electrical transport property evolution of the single nanowire under charging/discharging test has been reported for the first time," Liqiang Mai tells Nanowerk. "By designing single nanowire electrode devices, our findings show that conductivity of the nanowire electrode decreased reversibly – for vanadium oxide nanowire by shallow discharge/charge – or irreversibly – for vanadium oxide nanowire by deep discharge/charge, or silicon nanowire – during the electrochemical reaction, which limits the cycle life of the devices." | |
 |
|
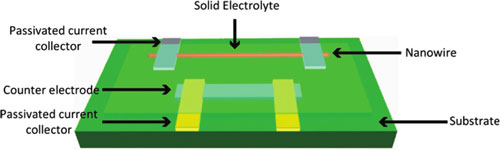
| Schematic diagram of a single nanowire electrode device design. A single vanadium oxide nanowire or silicon nanowire is the work electrode, and HOPG or LiCoO2 nanofilm is the counter electrode. The electrolyte is the PEO-LiClO4-PC-EC polymer. (Reprinted with permission from American Chemical Society) | |
| This first all-solid-state single nanowire electrochemical device, designed by Mai, a professor at the State Key Laboratory of Advanced Technology for Materials Synthesis and Processing at Wuhan University of Technology (WUT), and advanced research scholar at the Lieber Research Group at Harvard University, together with Yajie Dong, a graduate student of the same Harvard group, and their collaborators from the WUT-Harvard Joint Nano Key Lab, is a unique and versatile platform for in situ probing the intrinsic mechanism for electrode capacity fading, which is one of the biggest challenges in Li ion based energy storage devices. | |
| "The insight gained from our study could help understand why the capacities of lithium ion batteries fade during their life time and provide scientific basis for designing, diagnosing and optimizing high performance lithium ion based energy storage devices" says Dong. | |
| Restraining the conductivity decrease of battery electrode materials is a key issue for improving the performance of lithium ion batteries. The team has reported on their work on using chemical prelithiation to improve cycling performance of nanostructured electrode materials in a previous publication ("Improved cycling stability of nanostructured electrode materials enabled by prelithiation"). | |
| The electrode device in this research was configured with one vanadium oxide nanowire with a length of 10-45 µm and a diameter of 20-100 nm as cathode, and one flake of highly ordered pyrolytic graphite as anode. | |
| Dong explains that during battery charge or discharge, Li+ ions move out or into the cathode materials, oxidizing or reducing it to different states. | |
| "This process was usually studied ex situ after disassembling the battery. So far, only in situ XRD or NMR could provide some indirect hint on materials structure changes during the battery test. Our single nanowire battery design provides a unique advantage to study this in situ without disturbing the battery components." | |
| In addition, such a single nanowire electrochemical device could also find applications in providing the potential power needs of nanodevices in future self-powered nanosystems. Designed as a nanoscale electrical energy storage device, it could be combined with nanowire-based solar cells, nanogenerators, etc for powering nanodevices in the fields of nanoelectronics, optoelectronics or biosensing. | |
| Mai notes that the research and development of lithium ion based energy storage devices has been focused in two directions. | |
| "On one hand, in the areas of electric vehicles and other large grid scale energy storage devices, these batteries are becoming bigger and bigger. On the other hand, in the areas of microelectronics or even self-powered nanosystems, they are becoming smaller and smaller. In either direction, when the size scales start to diverge from traditional batteries by orders of magnitude, the old battery design won't be efficient enough to achieve high energy, power density and long cycle life with satisfactory safety features." | |
| "Novel efficient design based on deep understanding of battery behaviors, such as the intrinsic reasons of energy storage device performance degradation studied in our work, will be important for tomorrow's energy storage devices" Mai concludes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
