| Posted: Jan 18, 2011 | |
Size effect and vacancies in nanomaterials |
|
| (Nanowerk Spotlight) At the nanoscale, the properties of materials – mechanical, electrical, thermal, optical – often differ significantly from their bulk behavior. And while nanostructured and nanoengineered products are appearing in the marketplace, researchers are still trying to understand all aspects of materials properties of nanostructures and how they can be modified and controlled. | |
| Vacancies (also called Schottky defect) play a major role in the electrical and thermal transport as well as the mechanical behavior of materials. A vacancy is the simplest defect which can be created in a material – it corresponds to a lack of an atom in the lattice. | |
| New theoretical work by Dr. Grégory Guisbiers at the Université catholique de Louvain in Belgium calculates the size effect on the vacancy formation energy, the vacancy formation entropy and the vacancy concentration into nanomaterials through a top-down approach by using classical thermodynamics. | |
| Reporting his work in the January 20, 2011 online edition of Journal of Physical Chemistry C ("Schottky Defects in Nanoparticles"), Guisbiers' main findings is that the vacancy concentration increases when size reduces and temperature increases. This result is important because it helps to understand how the mechanical, electrical and thermal properties are modified at the nanoscale. The model considers the size effect on the vacancy formation energy and the vacancy formation entropy. The previous model from Qi et al. ("Surface-area-difference model for thermodynamic properties of metallic nanocrystals") only considered the size effect on the vacancy formation energy. Guisbier's model goes one step further. | |
| "To understand the processes occurring in nanomaterials during heat treatment and mechanical deformation, the size effects on the vacancy formation energy and entropy have to be considered," Guisbiers explains to Nanowerk. "The new model explains why nanomaterials appear to be perfect – it is due to the limited number of atoms in a particle (nanoparticles generally have less than one million atoms and according to the theory there are less than 1 vacancy per one million atoms)." | |
 |
|
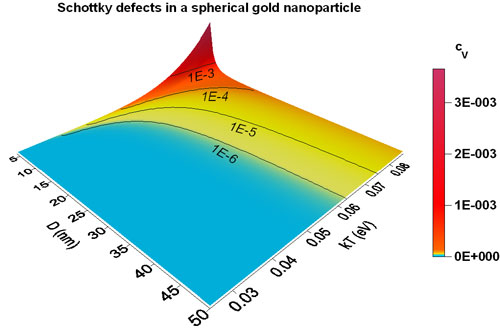
| The vacancy concentration of a spherical gold nanoparticle is plotted versus its size and temperature. The vacancy concentration increases dramatically (∼10-3) for small sizes (∼5nm) and high temperatures (T ∼1000K). (Image: Grégory Guisbiers) | |
| This model can also explain how defects like vacancies influence the properties of a material. It allows the calculation of the vacancy concentration, the vacancy formation energy, and the vacancy formation entropy for a nanoparticle. | |
| "Predicting the vacancy concentration in nanostructures is important because it can explain the mechanical, electrical and thermal properties observed in these nanostructures" says Guisbiers. "Indeed, the presence of vacancies in the crystal lattice modifies the lattice structure around the vacancies and then results in a decrease of the lattice parameter and a lattice softening. Furthermore, the hardness and the yield strength of the material typically increase with decreasing size, a phenomenon known as the Hall-Petch effect. The size-dependent hardness enhancement at a temperature well below the melting temperature is due to the bond length contraction and the associated bond strength gain. Increasing vacancy concentration results in a decrease of the electrical and thermal conductivity due to the scattering of electrons on vacancies and the lattice softening." | |
| The next stages in Guisbiers' investigations are to focus on interstitial defects and dislocations. The goal is to see how these defects can influence the nanomaterials properties. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
