| Posted: Jan 21, 2011 | |
Nanomaterials for next-generation ultrafast lithium storage |
|
| (Nanowerk Spotlight) Nanomaterials are key in developing high-performance energy storage technologies for mobile phone batteries and notebooks providing long battery times. Lithium-ion batteries are not only used in mobile gadgets but are currently being intensively developed worldwide for use in electric vehicles. As lithium-ion batteries are charged, large amounts of lithium ions are held in the anode. When the battery is used, these ions migrate to the cathode, sending electrons through the circuit. Traditional anode materials like graphite have a fairly low storage capacity and release rate, so finding alternatives is key to making batteries that last longer and produce more power. | |
| Titanium dioxide is regarded as one of the ideal candidates for high-rate anode materials, owing not only to its structural characteristics and special surface activity, but also to its low cost, safety, and relatively low environmental impact. | |
| Researchers in Singapore have developed a facile system to fabricate sandwich-like carbon-supported stacked titanium dioxide nanosheets, in which carbon pillars create open channels for fast lithium ion diffusion and the ultrathin framework renders the storage of lithium almost exclusively on the surface. This work provides a new route to design the electrode materials for quick-charging lithium ion batteries. | |
 |
|
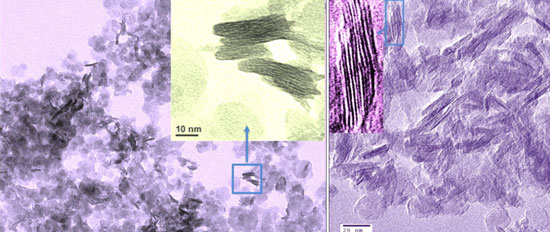
| TEM images of the as-synthesized layered titanate nanosheets (left) and the carbon-supported stacked titanium dioxide nanosheets obtained after annealing at 350 °C for 2 hours (right). (Source: Xuewei Liu, Nanyang Technological University) | |
| "The lack of open channels in bulk titanium dioxide is the main drawback that restricts its capacity and rate capability for reversible lithium insertion and extraction," Xuewei Liu, an assistant professor at Nanyang Technological University (NTU), explains to Nanowerk. "A reduction in the effective size and construction of open channels in the material are the main strategies currently employed to increase the rate performance. The capacity of ultrafine TiO2 nanocrystals and nanotubes, for example, is significantly enhanced at lower rates. However, their capacity and cycle life deteriorate dramatically at higher rates." | |
| Liu notes that, in this respect, significant efforts have recently been made on the fabrication of anatase TiO2 nanosheets with exposed highly reactive (001) facets. These TiO2 nanosheets are shown to be an excellent host structure for lithium insertion and extraction due to the presence of exposed (001) facets and short path along the [001] direction for lithium ion diffusion. | |
| "However, high-pressure systems including flammable solvent and corrosive additives (bases or acids) were often employed in solvothermal methods" says Liu. "There is still potential danger in the high-temperature and high-pressure systems when low boiling-point inflammable solvents are used. In addition, the nanosheets with exposed smooth facets easily overlap with each other because of the Ostwald ripening process, which leads to a decrease of surface area. To conquer these obstacles, we designed our simple ionic-liquid system." | |
| Reporting their findings in a recent issue of Advanced Materials ("Sandwich-Like, Stacked Ultrathin Titanate Nanosheets for Ultrafast Lithium Storage"), Liu's group, working with Lou Xiong Wen's team at NTU's College of Engineering, synthesized sandwich-like carbon-supported stacked TiO2 nanosheets (CTNSs) on a large scale by a facile and clean route. | |
 |
|
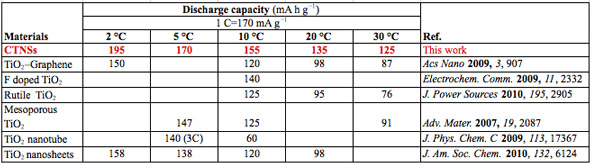
| Comparison with existing methods and materials (1 C = 170 mA g-1) (Source: Xuewei Liu, Nanyang Technological University) | |
| Owing to the unique textural features, i.e., stacked nanosheets with open channels, the storage of lithium in these CTNSs mainly takes place on surface, which is completely different from the insertion of lithium into bulk anatase TiO2 materials. | |
| "This pseudocapacitive insertion and extraction renders remarkable improvement in high-rate charging and discharging" says Liu. "Moreover, the in situ derived carbon can significantly stabilize these stacked TiO2 nanosheets, leading to an excellent cycle life. We thus anticipate the promising use of this material in high-power lithium-ion batteries." | |
| Liu summarizes the advantages of the new anode material: 1) Clean synthetic systems: Low pressure and safe; 2) The ionic liquid system is low-cost and facile to be synthesized; 3) High yield: the product could be synthesized in large scale and easy to mass-production; 4) Unique structure with superior performance: The sandwich structure provides open channels that are very suitable for fast lithium storage. The table above shows that the material has excellent capacities at low rates and high rates. The full charging time at 30 °C is only ∼1.3 minutes. | |
| Since the development of ultrafast-charging and high-rate electrode materials with low-cost is still a big challenge for next-generation lithium ion batteries, the NTU team will continue to focus their research on the development of novel and low-cost electrode materials for next-generation lithium ion batteries. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
