| Posted: Apr 14, 2011 | |
Nanoporous quantum filters - inside the fascinating and weird world of confined quantum particles |
|
| (Nanowerk Spotlight) One of the problems in modern separation science and technology is the challenge of separating gaseous mixtures that consist of very similar particles, for example, hydrogen isotope mixtures; mixtures of noble gases; etc. The problem arises because small particles such as hydrogen isotopes share similar size and shape (only their molecular mass is different). Moreover, these particles are usually very stable. While this problem can be technically solved, currently available separation methods such as thermal diffusion, cryogenic distillation, and centrifugation, tend to be time and energy intensive. | |
| New theoretical work now shows that narrow carbon nanotubes (CNTs) seem to be an attractive alternative. By using CNTs as nanoporous molecular sieves, the separation of parahydrogen molecules from mixtures of classical particles at cryogenic temperatures seems to be possible. | |
| In an upcoming paper in Physical Chemistry Chemical Physics ("Quantum fluctuations increase the self-diffusive motion of para-hydrogen in narrow carbon nanotubes", doi: 10.1039/c1cp20184k), a team led by Piotr Kowalczyk from the Nanochemistry Research Institute at Curtin University of Technology in Australia, is shedding new light on the separation of light particles, such as hydrogen isotope mixtures, via quantum molecular sieving. | |
| "We found that strong confinement of hydrogen molecules inside very narrow carbon nanotubes at cryogenic operating temperatures affects their self-diffusive motion significantly," Kowalczyk tells Nanowerk. "As one would expect, hydrogen molecules treated classically undergo highly correlated movement when their collision diameter approach carbon nanotube size – i.e., anomalous diffusion in quasi-one dimensional pores". | |
| He explains that this simply means that the self-diffusive motion of these classical particles is very slow inside narrow carbon nanotubes. | |
| But what about hydrogen molecules treated quantum mechanically? Here the story appears to be completely different thanks to the weird world of quantum mechanics. | |
 |
|
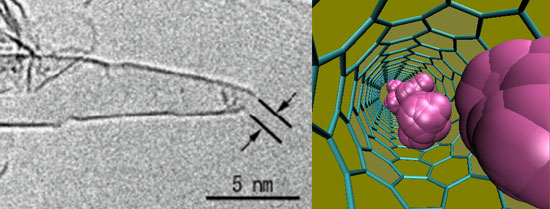
| Left: Single-walled carbon nanohorns discovered by Iijima et al. in 1999. High-resolution transmission electron microscopy image was taken by Dr Tanaka from Katsumi Kaneko's laboratory. Right: Self-diffusive motion of para-hydrogen molecules adsorbed in (6,6) carbon nanotube at 30 Kelvin. (Image: Dr. Kowalczyk, Curtin University of Technology) | |
| "By using Feynman's treatment of quantum mechanics at finite temperatures via path integrals, we found that quantum effects – more precisely, zero-point motion and tunneling – at cryogenic temperatures enhances self-diffusive motion of confined hydrogen molecules significantly" explains Kowalczyk. "This means that quantum particles self-diffuse inside carbon nanotubes even when the operating temperature is very low." | |
| How to explain this nontrivial theoretical prediction? | |
| What Kowalczyk and his collaborators from the Physicochemistry of Carbon Materials Research Group at Nicolaus Copernicus University in Poland did, was to combine classical Newtonian dynamics and Rabani-Reichman-Krilov-Berne treatment of quantum dynamics at finite temperatures (so-called the numerical analytic continuation method), with Feynman's path integral method, to provide a detailed microscopic picture of hydrogen self-diffusion in narrow carbon nanotubes. | |
| "We find that zero-point energy and tunneling significantly smooth out the free energy landscape of hydrogen molecules adsorbed inside a narrow (6,6) carbon nanotube" says Kowalczyk. "This promotes a delocalization of confined hydrogen molecules at the studied temperature of 30 Kelvin – i.e., quantum fluctuations impact their dynamics." | |
| What this means, in other words, is that hydrogen molecules treated quantum mechanically have more space for their self-diffusive motion than their classical counterparts (see movie below). | |
| Snapshots of molecular hydrogen adsorbed in a (6,6) carbon nanotube at 30 Kelvin | |
| A particularly interesting finding of this work is that hydrogen molecules adsorbed inside narrow carbon nanotubes are very "hot" particles. | |
| When calculating the exact kinetic energy of hydrogen molecules adsorbed in the studied carbon nanotubes, the team found that the kinetic energy of the adsorbed hydrogen molecules approaches 140 Kelvin at the studied temperature of 30 Kelvin. Note that at the same time the kinetic energy of hydrogen molecules treated classically is 45 Kelvin. | |
| "This is a consequence of the famous Heisenberg uncertainty principle" says Kowalczyk. "Once you localize the particle in space, its momentum has to increase." | |
| The practical conclusion from these theoretical results, according to the team, is that homogenous narrow carbon nanotubes are promising nanomaterials for the construction of novel filtration devices, for instance nanofilters with the ability to separate hydrogen molecules from mixtures of classical particles at cryogenic operating conditions. | |
| "Although our theoretical calculations shed new light on the problem of quantum dynamics of light particles in strong nanoconfinement at cryogenic temperatures, more theoretical and experimental work is needed to fully explore the potential of fascinating novel nanomaterials in separation science and technology" concludes Kowalczyk. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
