| Posted: Apr 20, 2011 | |
Life cycle assessment shows high energy requirements for fullerene production |
|
| (Nanowerk Spotlight) There is a general perception that nanotechnologies will have a significant impact on developing 'green' and 'clean' technologies with considerable environmental benefits. Just take a look at the dozens of Nanowerk Spotlights we have written about the use of nanotechnology in areas ranging from water treatment to energy breakthroughs and hydrogen applications. As a matter of fact, renewable energy applications probably are the areas where nanotechnology will make its first large-scale commercial breakthroughs (see: "Nanotechnology applications could provide the required energy breakthroughs"). | |
| In two previous Spotlights (here and here) we wrote about the fact that the environmental footprint created by today's nanomanufacturing technologies are conflicting with the general perception that nanotechnology environmentally benign. It actually appears that certain nanomaterial production technologies are quite dirty and also have a considerable energy footprint. | |
| Determining the full environmental impact of nanomaterials requires a full life cycle assessment. A recent paper in Environmental Science & Technology takes a look at the material and energy intensity of fullerene production, something that hadn't been done before. | |
 |
|
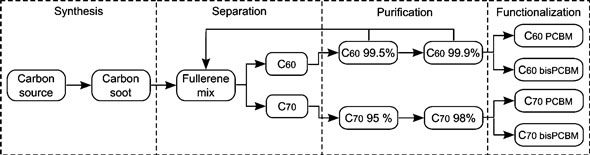
| Overview of the process flow for the production of modified fullerene compounds for use as functional materials in organic solar cells. (Reprinted with permission from American Chemical Society) | |
| As strong electron acceptors, fullerenes are promising materials for use in photovoltaics. They are already in commercial use, for instance in Konarka's organic photovoltaic technology. In this new paper, the authors, led by Brian J. Landi from Rochester Institute of Technology's NanoPower Research Labs and first authored by Annick Anctil, conduct a life cycle assessment (LCA) to assess energy intensity of producing C60 and C70 fullerenes and corresponding derivatives that are being considered in present day medical and energy applications. With their analysis of the various fullerenes products, the authors intend to provide a baseline to evaluate future product developments and guide product design and research. | |
| According to the team, "whereas previous studies have limited the LCA scope to direct energy input – e.g., electricity usage – during fullerene synthesis, this study quantifies total material intensity and embodied energy with a scope that includes all direct and upstream feedstock and fuel energy inputs for modern production methods". | |
| "The primary goal of this study is to use the calculated energy intensity of modified fullerene production to determine opportunities for future process innovations to minimize environmental impact," says Anctil. "As secondary objectives, this study serves both to fully document a life cycle inventory of fullerene production and to investigate the relative importance of nanomaterials in the products in which they are used, as demonstrated through examples of C60 used in an aluminum composite and PCBM derivative used in organic solar cell applications." | |
| In contrast to previous work, the new LCA quantifies embodied energy inclusive of all upstream inputs, not just direct process energy for plasma or embodied energy of the carbon feedstock for pyrolysis synthesis techniques. It also takes into account production methods published in the last five years. | |
| Altogether, the embodied energy of fullerene production has to take into account synthesis, separation, purification and functionalization steps. | |
| While synthesis and separation are only the first steps of functional fullerene production, purification is an important step to meet the higher purity requirements demanded by commercial applications. Therefore, the true embodied energy in a product is related to the solvents, chemicals, and electricity required to isolate the purified product. | |
| In addition to the high-purity requirements, certain applications also require a modification of the fullerene structure. The embodied energy associated with the various chemicals, solvents, and solvent regeneration steps necessary for this functionalization further increase the overall amount of energy inputs. These steps also lead to material losses that require additional raw materials. | |
 |
|
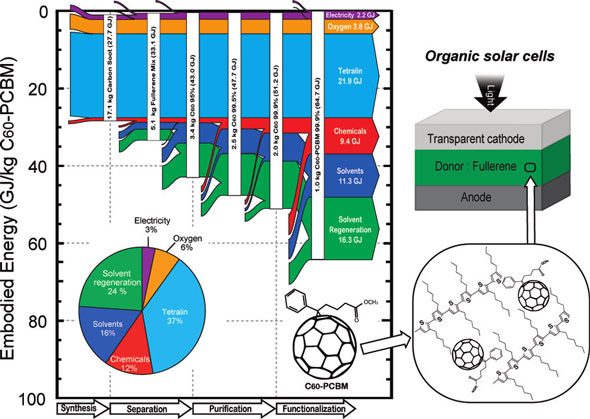
| Energy flow diagram for production using the pyro-tetralin synthesis method of 1 kg of C60-PCBM. (Reprinted with permission from American Chemical Society) | |
| The authors point out that the embodied energy of all fullerenes are an order of magnitude higher than most common chemicals and, therefore, are likely to influence the embodied energy of the product they will be used in, even though they might only represent a small fraction of its total mass. | |
| "For a typical organic solar cell (P3HT:PCBM), the fullerene would only account for 0.3% of the total weight according to previous work and would be excluded based upon recent reports using mass based cutoff rule" write the authors. "However, in the case of the active layer for an organic solar cell, the addition of C60-PCBM 99.9% (pyro-tetralin) to the polyhexylthiophene will increase the polymer embodied energy from 160 MJ/kg to 32,400 MJ/kg. Scaling this increase up to the level of the full device, fullerenes would then represent up to 19% of the total embodied energy of the solar cell." | |
| The team concludes that, since it is clear that the high embodied energy of nanomaterials will have a significant impact on the final product embodied energy, cutoff rules based on mass should not be applied routinely to simplify lifecycle assessment of products containing nanomaterials. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
