| Posted: Jun 28, 2011 | |
Nanotechnology-enabled fuel cells could make coal power significantly less dirty |
|
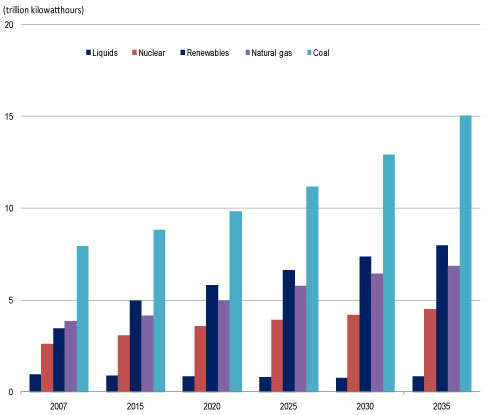
| (Nanowerk Spotlight) Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide – 79% in China, 69% in India, 49% in the USA, and 46% in Germany (source). For the foreseeable future, coal will continue to be the dominant fuel for electric power generation. | |
 |
|
| World net electricity generation by fuel, 2007-2035. Derived from EIA, International Energy Statistics database (as of November 2009). Projections: EIA, World Energy Projection System Plus (2010). | |
| However, the conventional process of converting coal into electricity has several severe drawbacks: | |
| First, the energy efficiency is low – today's plants convert only a third of coal's energy potential to electricity. | |
| Second, coal mining and burning raises a number of massive environmental challenges, including soil erosion, dust, impacts on local biodiversity, air and water pollution. Coal fired power plants are the biggest source of man made carbon dioxide emissions. Burning coal is a leading cause of smog, acid rain, global warming, and air pollutants. In an average year, a typical coal plant generates: | |
|
|
|
| Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Compared to traditional technologies, SOFCs have a higher efficiency, lower noise and lower emissions of, e.g., carbon dioxide. They work by transforming the chemical energy of a fuel directly into electricity. | |
| Especially gasified carbon fuel cells (GCFCs) offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. | |
| GCFCs are twice as efficient as today's coal-fired power plants, potentially offering a way to cut CO2 emissions in half. Another benefit of their use is that the effluent stream of almost pure CO2 from GCFCs is ideally suited for direct sequestration or industrial use without the need for CO2 separation. | |
| "To date, the performance and stability of GCFCs are still inadequate for practical applications, due primarily to insufficient activity of the anodes," Meilin Liu, a Regents professor in the School of Materials Science and Engineering at the Georgia Institute of Technology, tells Nanowerk. "Furthermore, the existing fuel cell anodes are vulnerable to coking by dehydrogenation of hydrocarbons and CO disproportionation at temperatures below 850°C, where SOFCs become more competitive economically." | |
| In new work, reported in the June 21, 2011 online edition of Nature Communications ("Promotion of water-mediated carbon removal by nanostructured barium oxide/nickel interfaces in solid oxide fuel cells") and first-authored by Lei Yang, a team led by Liu developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius. The team included researchers from Brookhaven National Laboratory, New Jersey Institute of Technology, and Oak Ridge National Laboratory. | |
| SOFCs rely on anodes made from nickel and a ceramic material known as yttria-stabilized zirconia (YSZ). Until now, however, carbon-containing fuels such as coal gas or propane could quickly deactivate these anodes, clogging them with carbon deposits in a process known as coking – especially at lower operating temperatures. | |
| To counter this problem, Liu's team have developed a technique for growing barium oxide nanostructures on the anodes. The structures adsorb moisture to initiate a water-based chemical reaction that oxidizes the carbon as it forms, keeping the nickel electrode surfaces clean even when carbon-containing fuels are used at low temperatures. | |
| This technique uses a vapor deposition process to apply barium oxide nanoparticles to the Ni-YSZ electrode. The particles, which range in size from 10 to 100 nanometers, form "islands" on the nickel that do not block the flow of electrons across the electrode surface. | |
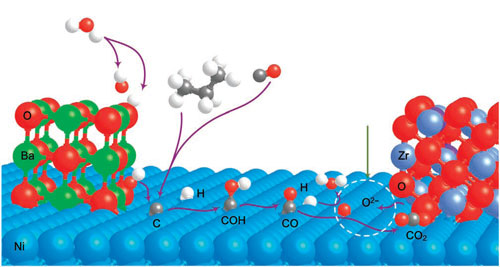
| When water vapor introduced into the coal gas stream contacts the barium oxide, it is adsorbed and dissociates into protons and hydroxide (OH) ions. The hydroxide ions move to the nickel surface, where they combine with the carbon atoms being deposited there, forming the intermediate COH. The COH then dissociates into carbon monoxide and hydrogen, which are oxidized to power the fuel cell, ultimately producing carbon dioxide and water. About half of the carbon dioxide is then recirculated back to gasify the coal to coal gas to continue the process. | |
 |
|
| Proposed mechanism for water-mediated carbon removal on the anode with BaO/Ni interfaces. Large balls in Brandeis blue, green, red, blue grey and purple are Ni, Ba, O of BaO or YSZ, Zr and Y, respectively, whereas small balls in red, white and grey are O from H2O, H and C, respectively. D1 is the dissociative adsorption of H2O, whereas D2 is the dehydrogenation of hydrocarbons or the CO disproportionation reaction. (Reprinted with permission from Macmillan Publishers) | |
| "While a number of alternative anodes have been investigated, the application of them to actual fuel cell systems is hindered by several critical issues," explains Liu: "They are too expensive to be economically viable – e.g., using noble metal such as ruthenium or palladium – or incompatible with current SOFC systems. It has been difficult to adopt alternative anode materials in state-of-the-art fuel cell systems based on YSZ electrolytes, which have evolved progressively in the past few decades. The difficulties originate largely from alternative anodes' limited physical, chemical, and thermal compatibility with YSZ electrolyte during fabrication at high temperatures." | |
| By contrast, the anodes produced in Liu's technique are compatible with standard solid oxide fuel cell systems that are already being developed for commercial electricity generation, home power generation and automotive applications. The barrier for implementing it in conventional fuel cell systems should be quite low. | |
| "Because our electrode builds on existing technology, there is a lower barrier for implementing it in state-of-the-art fuel cell systems" he says. | |
| In addition to use in gasified coal fuels, this novel anode could potentially employ a wide variety of carbonaceous solid fuels such as municipal solid waste and biomass, representing a new generation of SOFCs for efficient conversion of readily available fuels to electricity with minimal emissions. | |
| "This could ultimately be the cleanest, most efficient and cost-effective way of converting coal into electricity," says Liu. "Meanwhile, it could produce the effluent gas with much purer carbon dioxide than the stack gases leaving traditional coal-fired power plants. That would make capturing the carbon dioxide for sequestration less expensive by eliminating large-scale separation and purification steps." | |
| He notes that, so far, the team has conducted the operation of gasified coal fuel cells for as long as hundred hours, and saw no evidence of carbon build-up. A major challenge ahead is to test the long-term durability of the system for fuel cells that are designed to operate for as long as five years. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
