| Posted: Jul 24, 2007 | |
Plugging into the brain with carbon nanofibers |
|
| (Nanowerk Spotlight) Just a few days ago we ran a Spotlight on nanobionics that addressed some of the issues of bridging the interface between electronics and biology. Today we'll take a look at some leading edge research in the field of neural engineering – an emerging discipline that uses engineering techniques to investigate the function and manipulate the behavior of the central or peripheral nervous systems. | |
| Neural engineering is highly interdisciplinary and relies on expertise from computational neuroscience, experimental neuroscience, clinical neurology, electrical engineering and signal processing of living neural tissue, and encompasses elements from robotics, computer engineering, neural tissue engineering, materials science, and nanotechnology. | |
| In order for neural prostheses to augment or restore damaged or lost functions of the nervous system they need to be able to perform two main functions: stimulate the nervous system and record its activity. To do that, neural engineers have to gain a full understanding of the fundamental mechanisms and subtleties of cell-to-cell signaling via synaptic transmission, and then develop the technologies to replicate these mechanisms with artificial devices and interface them to the neural system at the cellular level. The first steps toward precise, informative and biocompatible neural interfaces have been made already. | |
| Scientists' understanding of distributed neuronal network processing has been facilitated by the advent of microelectrode array (MEA) recording platforms, capable of recording activity from multiple neurons simultaneously. | |
| "Simultaneous recording from multiple locations within a tissue may unlock the neural code underlying higher brain functions" Dr. Barclay Morrison explains to Nanowerk. "We now have introduced a new type of MEA with electrodes made not of metal but of vertically aligned carbon nanofibers (VACNFs). We have shown that these arrays can perform the standard complement of electrophysiological techniques possible with commercial MEAs." | |
| Researchers have already demonstrated the potential of nanofiber-based electrode architectures for interfacing with excitable cell matrixes ("Resident Neuroelectrochemical Interfacing Using Carbon Nanofiber Arrays"). Carbon nanofibers are electrochemically active structures that may be integrated into parallel arrays using the conventional tools and approaches of microfabrication. In contrast to conventional planar arrays, however, nanofibers provide a novel, nonplanar, high aspect ratio structure that may provide unique opportunities for probing extra-, inter-, and ultimately intracellular phenomena. | |
  |
Images are of dissociated cultures on VACNF arrays. Red cells are astrocytes and green are neurons. Blue dots are nuclei. Left: The central portion of the VACNF array - the line running horizontally - contains the CNF electrodes. The cells were isolated from a P8 rat pup cortex, dissociated and plated on the array. They were grown for about a week before being fixed and then stained for cell specific markers: red is GFAP which is for astrocytes and green is for b3 tubulin which is for neurons. The blue is DAPI which stains all nuclei. Basically, the experiment showed that the arrays were biocompatible and that brain cells could survive on the structure. Magnification is 200X; Right: This image is of a different chip. Essentially the same culture preparation, but the neurons were purified more before plating. So the field of view has no astrocytes in it. The CNF electrodes can be seen as the black dots at the end of the metal connectors in the substrate. Magnification is 400x. (Images: Dr. Morrison, Columbia University) |
| "We have previously demonstrated that these structures could be interfaced to individual cells, but it was not known if they would be robust enough to interface to intact tissue" says Morrison. "Further, it was not known whether our carbon nanofibers could stimulate and record action potentials from neural tissue. We have shown that both were possible with our vertically aligned carbon nanofiber arrays." | |
| Morrison, Assistant Professor in Biomedical Engineering at Columbia University, and collaborators from his department and the Oak Ridge National Laboratory, led by Timothy E. McKnight and Milton Nance Ericson, and were able to demonstrate that these same carbon nanofiber electrodes may also be used to stimulate and record electrophysiological signals from intact hippocampal slices ("Vertically Aligned Carbon Nanofiber Arrays Record Electrophysiological Signals from Hippocampal Slices"). | |
| For their experiments, the researchers fabricated arrays which consisted of a linear array of 40, individually addressed VACNF electrodes, 10 µm in height, spaced 15 µm apart along a total length of 600 µm. To record electrical activity, a slice of live hippocampal tissue was positioned on the VACNF array, allowing the electrodes to penetrate into the tissue. Morrison and his colleagues were able to record spontaneous activity as well as responses evoked by stimuli applied to the electrodes. | |
| In the field of neural engineering, carbon electrodes have several potential advantages over other electrode materials. Morrison explains that, perhaps most importantly, carbon electrodes are well suited for electrochemical measurements in neuronal environments and have been used to observe dynamic chemical events in and around neuronal tissue. "Combined with their ability to also record electrophysiological signals, this dual sensing capability opens up the door to developing neural prostheses that sense both neurotransmitter levels and electrical signals simultaneously" he says. | |
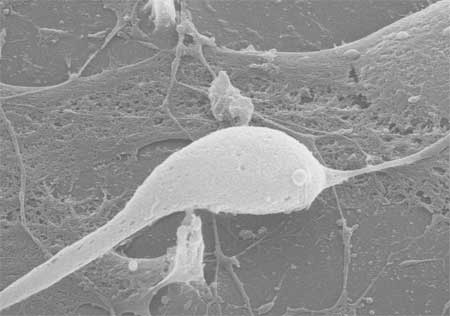 |
SEM image of a cell, most likely a neuron, interacting with a single VACNF. The cell has one main process and one minor process coming off the cell body. The cell body is in close proximity to an electrode (the light vertical structure). One can also see either extra cellular matrix or thin glial cells (astrocytes) around the cell in the center. Some of this material has been attached to the electrode at the bottom of the image. (Image: Dr. Morrison, Columbia University) |
| The long-term goal of this research is to demonstrate the ability to measure both electrophysiological signals and neurotransmitter concentrations using these novel VACNF microelectrode arrays. Morrison hopes that this ability could ultimately lead to improved neural prosthetics that can interface to tissue with high numbers of connections, while also providing feedback on the local chemical environment, including tissue health or activity over time. | |
| In addition to future neuroprosthetic platforms, there are many applications within the neuroscience and neural engineering fields. "Dual sensing micro electrode arrays could increase our fundamental understanding of neuronal network dynamics – how groups of neurons communicate with each other – and how the network gives rise to emergent properties that underlie how the brain functions" says Morrison. "The dual sensing electrodes would provide information about not only the output of the neuronal network (electrical signals) but also the inputs (local neurotransmitter concentrations) that drive the electrical activity." | |
| This dual sensing ability could find use in neural prostheses, as well. Local neurotransmitter concentration could serve as a feedback channel for devices like deep brain stimulators so that their output could be modulated with respect to the brain's response. | |
| Although carbon-based electrodes may be functionalized with enzymes to provide analyte specificity to other less easily oxidized species, functionalization of the electrodes for the measurement of difficult-to-oxidize neurotransmitters will be a challenge. Morrison also points out that miniaturization of active circuitry beneath each electrode for amplification and electroanalytical measurements will be a challenge but should significantly improve signal to noise ratio as well as ease of use with a lab-on-a-chip platform. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
