| Posted: Sep 09, 2011 | |
High content screening of zebrafish greatly speeds up nanoparticle hazard assessment |
|
| (Nanowerk Spotlight) With the mass production of engineered nanoparticles, risk assessment efforts are in need of platforms that offer predictive value to human health and environment, and also possess high throughput screening capacity. Scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish (Danio rerio). Zebrafish share the same set of genes as humans and have similar drug target sites for treating human diseases. They are small, easy to maintain, and well-suited for whole animal studies. Furthermore, their early embryonic development is completed rapidly within five days with well-characterized developmental stages. The embryos are transparent and develop outside of their mothers, permitting direct visual detection of pathological embryonic death, mal-development phenotypes, and study of real-time transport and effects of nanoparticles in vivo (see for instance: "Nanoparticles' random walk has implications for nanotoxicology"). | |
| However, the current screening process in zebrafish involves mostly counting the survival rate, hatching and developmental abnormalities etc. through visual examination of each embryo and/or larvae under a dissecting microscope. Such process is time-consuming, labor-intensive and has limitations on data acquisition as well as statistics analysis. Researchers say that advanced imaging technology and automated testing are key challenges toward developing high content screening of nanomaterials, chemicals, and drugs in zebrafish. | |
| "With the development of high content imaging, one can acquire hundreds to thousands of microscopic images with high throughput and the images would allow further in silico image analysis and hazard ranking tools for engineered nanoparticles to greatly speed up the screening process," Sijie Lin, a postdoctoral researcher at the University of California's Center for Environmental Implications of Nanotechnology (CEIN), tells Nanowerk. | |
| Lin is first author of a paper in the August 18, 2011 online edition of ACS Nano ("High Content Screening in Zebrafish Speeds up Hazard Ranking of Transition Metal Oxide Nanoparticles") where an international team of collaborators successfully demonstrated two high content imaging platforms to enhance the ability to screen the toxicological effects of nanoparticles in zebrafish embryos. | |
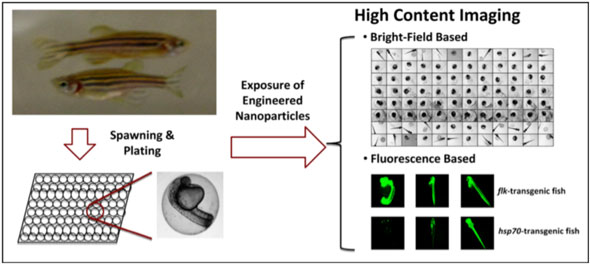
| "Our study provides, for the first time, the use of high content imaging – which includes bright-field and fluorescence based imaging – to compare the toxicological effects of transition metal oxide nanoparticles in zebrafish embryos and larvae," says Lin. "We have demonstrated the embryo hatching interference exerted by nanoparticles due to their intrinsic material characteristics, i.e. shedding of heavy metal ions, through bright-field high content imaging." | |
 |
|
| A schematic diagram that illustrates the procedure of high content screening in zebrafish. Embryos collected from healthy adult zebrafish fish was placed in 96-well microtiter plate where they received nanoparticles exposure. High content imaging (bright-field and fluorescence based) was conducted every 24 hour after exposure for 5 consecutive days. (Reprinted with permission from American Chemical Society) | |
| The team describes the use of an imaging platform for bright-field imaging analysis, capable of capturing phenotypic and developmental abnormalities in embryos and larvae. For larvae, the team also captured the stress induced by different types of nanoparticles on the larval stage of a transgenic zebrafish line (hsp70:eGFP) by fluorescence based high content imaging. | |
| "In order to apply these imaging tools to new engineered nanomaterials not previously assessed, we utilized a series of transition metal oxide nanoparticles – CuO, NiO, and Co3O4 – to compare to ZnO (positive control)" explains Lin. "Our results demonstrate profound interference in embryo hatching in parallel with hsp70:eGFP expression in larvae by CuO and NiO but not Co3O4." | |
| The researchers assume that the profound hatching interference and stimulation of hsp70 gene expression by CuO, ZnO, and NiO nanoparticles could be attributed to nanoparticle dissolution and shedding of transition metals. They were able to reverse these toxic effects with the metal ion chelator, DTPA. | |
| With this work, the team shows how high content image analysis can be used to perform hazard assessment in zebrafish in a time- and dose-dependent fashion. This approach can speed up the in vivo hazard assessment of engineered nanomaterials and may allow the development of an automated system that can also be used for structure activity analysis and hazard ranking. | |
| The researchers note that this platform can also be easily adapted for screenings other chemicals and/or drugs. | |
 |
|
| This image represents a 96-well microtiter plate used for the screening of nanoparticles in zebrafish. Each well of this plate was filled with one embryo and the bright-field based high content imaging automatically acquired one image per well. The images were then combined to show the screening results as well as layout of the plate. (Image: Sijie Lin CEIN) | |
| As more and more data is generated on the toxicological effects of engineered nanoparticles, scientists are no longer satisfied with identifying which nanoparticles are toxic, and to what degree, but they are also trying to boost the throughput capacity of their screening set-ups as well as understand the exact underlying toxicity mechanisms. The challenges that remain to be overcome are how to link the specific physicochemical properties of nanoparticles to their toxicological outcomes both in vitro and in vivo. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
