| Posted: Mar 19, 2012 | |
A precise nanothermometer for intracellular temperature mapping |
|
| (Nanowerk Spotlight) Green Fluorescent Protein (GFP) – originally found in a jellyfish – has played a crucial role in life science research, providing insights to many fundamental questions that have paved the way to the biology and medicine of the future. Since the mid-1990s, when the protein was successfully cloned, GFP can be found in research laboratories worldwide used as a visual marker of gene expression and protein localization, easily observed via light (optical) microscopy. GFP can be linked to other proteins and is primarily used to track dynamic changes in living cells. In 2008, biologists who discovered and developed the protein as a laboratory tool won a Nobel Prize for their work. | |
 |
|
| On this confocal microscopy image with fluorescent protein labeled details from the brain stem, tube-like structures are axons of the auditory pathway, which are forming hand-shaped synapses on other neurons (here unlabeled). (Confocal image by Jean Livet) You can find this and other stunning images on Mark Zimmer's Green Fluorescent Protein site. | |
| In new work, reported in the March 6, 2012 online edition of Nano Letters ("Mapping Intracellular Temperature Using Green Fluorescent Protein"), researchers in Spain have demonstrated how GFP can also act as an efficient nano-thermometer inside cells. | |
| Currently there is no reliable method to measure intracellular temperature. This novel approach is based on GFP, where the GFP is transfected by the cells. Using this approach, the cell mechanisms are not modified or altered during the measurement by the presence of GFP. | |
| "Because temperature governs many vital cellular processes, its accurate and non-invasive monitoring should contribute, in parallel with other existing GFP-based techniques, to further unravel the complex machinery of cell mechanics where the temperature plays an important role," Romain Quidant, ICREA Professor at The Institute of Photonic Sciences (IFCO), tells Nanowerk. | |
| Several microscopy techniques have recently been proposed to address the need for monitoring intracellular temperature in molecular biology. Most of them rely on introducing synthetic nano-objects into living cells, a fact that can cause stress in the cells and alter their behavior (see for instance our Nanowerk Spotlight: "How to build a nanothermometer"). | |
| Quidant and his team wanted to find a method to measure temperature that does not harm the cell, but at the same time can give robust imaging opportunities – namely, fast and accurate measurements. Their approach relies on measuring Fluorescence Polarization Anisotropy (FPA) of Green Fluorescent Protein and combines fast and accurate acquisition with confocal spatial resolution. | |
| Jon Donner, a PhD student in Quidant's group and a co-first author of the paper, explains the – well-established – underlying physics which relates temperature and molecular FPA: "In general, a population of fluorophores illuminated by a linearly polarized light re-emits partially polarized fluorescence due to the random orientation of the molecular dipoles. When the temperature increases, the Brownian rotational motion of the fluorophores is accelerated. Hence, the molecules will rotate more during their fluorescence lifetime. The more the molecules rotate during their fluorescence lifetime, the more the re-emitted photons will lose the memory of the incident light polarization. Consequently, a temperature increase leads to a decrease of the degree of polarization (anisotropy) of the fluorescence. Using a suitable calibration, FPA leads to an absolute temperature measurement. This is one of the main advantages of this technique." | |
| In their temperature measurement experiments, the researchers used single living cells and dispersed gold nanoparticles in the extracellular medium. Then they locally heated the cells by focusing an infrared laser 50 µm aside from the studied cell (they chose not to shine on the cell directly to demonstrate that FPA variations measured in the cytosol are unambiguously due to temperature variations and not to a possible IR-assisted perturbation of the GFP fluorescence emission process). They were able to precisely measure the temperature inside the cell resulting from the heated nanoparticles outside it. | |
 |
|
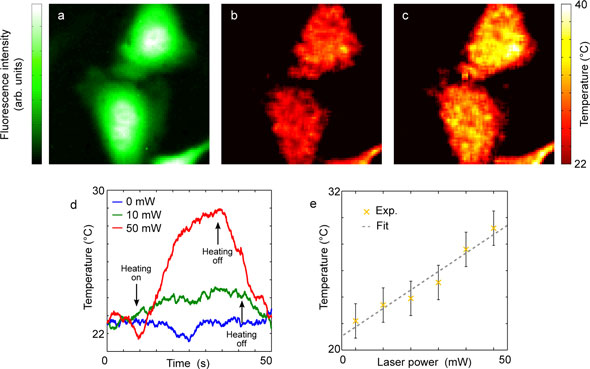
| Temperature measurements in HeLa cells while delivering local heat via photothermal approach. (a) Fluorescence intensity of GFP transfected HeLa cells. (b) Temperature map while not heating. (c) Temperature map while heating the NR with a focused infrared laser with a power of P = 50 mW, located 50 µm to the right of center of the image. (d) Temperature variation of a point within the cell as function of time. (e) Temperature of a point within a cell for different laser heating powers. (Reprinted with permission from American Chemical Society) | |
| "We hope that the method we are presenting here will become a powerful tool to unravel intimate cellular processes or mechanics at the single cell level," says Sebastian Thompson, a postdoc in Quidant's group who co-first authored the paper with Donner. "Benefiting from its full compatibility with widespread GFP cellular biology, it complements the existing toolbox for biologists and has the potential to extend our understanding of life science." | |
| He notes that this approach could become particularly useful for temperature-based therapies such as photothermal cancer therapy, where heat is locally delivered to cancer tumor cells via photothermal conversion of nanoparticles. This is a field that is already quite advanced and where clinical trials are being conducted. To better calibrate the parameters of these processes one needs to monitor the temperature of the cells by disturbing the cells as little as possible. | |
| Going forward, the researchers will work on improving the sensitivity of their method. | |
| "After that, we would love to see this research extended to in vivo applications" says Quidant. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
