| Posted: Mar 21, 2012 | |
Ultrasensitive chemical nanocoax sensor based on carbon nanotubes |
|
| (Nanowerk Spotlight) The ability to detect small quantities of analytes has been advanced by a number of independent approaches, many with nanotechnological features. Sensor devices built with nanotechnology go far beyond just the effort to miniaturize sensors by reducing the size of the sensing part and/or the transducer. Nanoscience deals with new physical or chemical properties of matter at the nanoscale, and new sensor devices are being built that take advantage of these phenomena. Important characteristics and quality parameters of the nanosensors can therefore be improved over the case of classically modeled sensors merely reduced in size. | |
| What is most desirable is a detection platform that facilitates rapid – as close to real time as possible – selective, and ultrasensitive detection of a number of targets, simultaneously. For low resource environments, the detection instrumentation must be low cost, low weight and low tech. | |
| Various types of nanostructures are used in the development of nanosensors: nanoparticles, nanotubes, nanorods, two-dimensional materials like graphene, embedded nanostructures, porous silicon, and self-assembled materials. For instance, gas sensors often operate by detecting the subtle changes that deposited gas molecules make in the way electricity moves through a surface layer. Researchers have fabricated gas sensors based on carbon nanotube-based field effect transistors (FETs), which can detect electrical potential changes around them (see our Nanowerk Spotlight: "Nanotechnology puts your nose on a carbon nanotube"). | |
| "While these and related sensing schemes can be all-electronic – i.e., not requiring optical readout – they all require sophisticated nanolithographic techniques to isolate, identify, and integrate electrical contact to the active nanosensor," Michael J. Naughton, Ferris Professor and Chairman of the Department of Physics at Boston College, explains to Nanowerk. "These structures are usually lying on a planar substrate, which can severely limit access of target molecules to the sensing element; fabrication of large numbers of these sensors can be costly and time-consuming; and temperatures of several hundred degrees centigrade and detection times of minutes are usually required to achieve the reported sensitivities." | |
| Reporting their work in the March 6, 2012 online edition of ACS Nano ("Ultrasensitive Chemical Detection Using a Nanocoax Sensor"), Naughton and his team, which included Drs. Dong Cai and lead author Huaizhou Zhao, have now presented a nanoscale 3D architecture that can afford highly sensitive, room temperature, rapid response, and all-electronic chemical detection. | |
 |
|
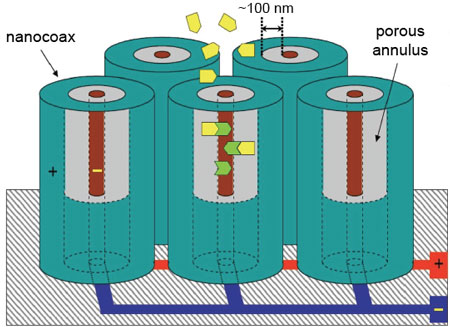
| Composed of an array of vertically aligned nanoscale coaxial electrodes constructed with porous dielectric coax annuli around carbon nanotube cores, this sensor is shown to achieve parts per billion level detection sensitivity, at room temperature, to a broad class of organic molecules. (Image: Dr. Naughton, Boston College) | |
| Inspired by the coaxial cable, derived from a recently elaborated nanocoax array architecture developed at Boston College for nanoscale manipulation of light and nanostructured solar cells (see for instance ""Efficient nanocoax-based solar cells""), the team adopted a nanocoaxial architecture and modified its structure to function as an array of coaxial capacitors. This nanocoax array technology uses relatively simple capacitance detection. | |
| "Having 100 million sensors per square centimeter, it is readily amenable to multiplexing," says Naughton. "As a chemical sensor, a porous coax annulus is employed, as shown in this work, to adsorb target molecules for detection at the parts per billion range, and below. This appears to be the most sensitive aluminum oxide-based chemical sensor." | |
| To fabricate the nanocoax, the researchers coated an array of vertically oriented carbon nanotubes with an aluminum oxide (Al2O3) dielectric and aluminum metal to form a coaxial electrode. | |
| Naughton points out that each unit in these arrays is in fact a coaxial capacitor. "Target molecules entering porous annuli of such open-ended nanocoaxial wires – e.g., via diffusion and chemical and/or physical adsorption – will cause changes in the dielectric response (capacitance and conductance) in proportion to their number and their static and dynamic dielectric properties." | |
| He notes that the nanoscale character of the coaxial electrodes is such that devices can be fabricated to be sensitive to the presence of very small numbers of molecules (i.e. of order 10) in under one minute. This exquisite sensitivity follows from the well-known ability to measure capacitance with high sensitivity. | |
| There are many potential applications for this novel sensing device. One concrete example is the analysis of volatile organic metabolites in breath for the early detection of human disease, such as cancer or diabetes. In addition, an increasing number of human diseases exhibit a biomarker signature in the blood comprised of macromolecules, e.g. proteins and glycolipids, well before conventional screens indicate the presence of disease. | |
| The Boston College team's device, properly functionalized for specific biomarker recognition, may provide the necessary sensitivity for detection of early disease states, such as cancer. | |
| "As a chemical detector, we perceive potential applications in environmental and defense-oriented sensing, as well as breadth-based analyte sensing," Naughton adds to the list of likely uses. "Also, we are presently further modifying the structure to form a biological sensor, binding linker molecules to the various nanocoax components to enable target-specific detection by the same capacitance method. This could be useful for a wide variety of targets, from pathogens to disease biomarkers, e.g. CA125 protein indicative of ovarian cancer. We see a range of applications from diagnostic to environmental sensing/detection." | |
| Looking further into the future, the prospect exists for a highly multiplexed, chip-based point-of-care / point-of-use molecular-scale biosensor / chemical sensor platform that operates off a portable power supply, such as a smart phone. According to Naughton, many research groups are thinking and working along these lines. | |
| "However, our approach does not employ or require lasers for target fluorescence or polymerase chain reaction for signal amplification, instead using simpler capacitance detection in a miniaturized format," he says. "Of course, there is a large number of challenges to any technology development such as this. Our first priority is getting the science right, by demonstrating highly specific detection with high reproducibility for a single biomolecular target. Our second is to demonstrate on-chip multiplex capability. Having success at these can lead to a rapid and progressive path forward to an inexpensive device appropriate for use in low resource environments, with the potential to positively impact numerous lives." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
