| Posted: Mar 28, 2012 | |
The promise of nitric oxide-releasing nanoparticles as wound healing agent |
|
| (Nanowerk Spotlight) Chronic, slow-healing wounds primarily affect elderly individuals and persons with conditions such as diabetes, peripheral vascular disease, and immune suppression. Older men and women with one or more of these conditions are more likely to experience and suffer from slow-healing wounds. Slow or non-healing wounds will often decrease their quality of life and impose significant financial stress. It is estimated that between 2.5 to 4.5 million people in the US are affected by these chronic, oftentimes disabling wounds. Furthermore, this chronic barrier disruption serves as a point of entry for pathogens, further impeding wound closure and quality of life. | |
| Wound healing is an exceedingly complex process, involving a multitude of signaling pathways, effector molecules, response phases, as well as a moderated balance between all these components. In certain disease states such as diabetes and HIV, this delicate balance is disturbed and wound healing is delayed, resulting in non-healing wound sites and therefore profound morbidity. | |
| Nitric oxide (NO) plays a critical role in the wound-healing process via antimicrobial properties, modulation of platelet/cytokine function, vasodilatory effects, and promotion of angiogenesis and matrix deposition. | |
| While attempts to administer NO to wound areas have shown some promise, the current modalities all suffer from varying drawbacks, such as administration site irritation or the burden of large, expensive equipment. | |
| "One commonly used NO donor, diethylenetriamine diazeniumdiolate (DETA NONOate), a type of diazeniumdiolates, has been shown to accelerate wound closure in rats," Adam Friedman, Director of Dermatologic Research and Associate Residency Program Director at the Unified Division of Dermatology of Montefiore Medical Center - Albert Einstein College of Medicine, tells Nanowerk. "However, clinical concerns regarding triamine-based DETA NONOates have been raised – they have been shown to migrate from the site of application prior to NO release, the soluble diamine and triamine byproducts of DETA NONOate are potentially toxic to tissue, and when introduced into the vasculature, they can cause methemoglobenmia." | |
| In a paper in the March 6, 2012 online edition of Nanomedicine: Nanotechnology, Biology and Medicine ("Nitric oxide-releasing nanoparticles accelerate wound healing in NOD-SCID mice"), Friedman and his team introduce a nanoparticle platform comprised of silane based sol-gel and sugar-derived glasses that can generate, store, and deliver NO in a controlled and sustained manner is utilized to enhance wound healing in immunodeficient mice. | |
 |
|
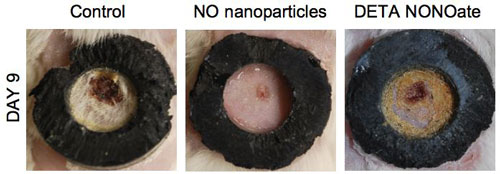
| Every other day dosing of a novel nitric oxide (NO) releasing nanoparticle platform applied to a splinted 5 mm punch biopsy full thickness wound in NOD-SCID mice resulted in accelerated wound closure as compared to controls, and as shown here on day 9, to comparable doses of a well known NO donor, DETA NONOate. (Image: Dr. Friedman, Albert Einstein College of Medicine) | |
| In addition to demonstrating promising antimicrobial activity, these nitric oxide-releasing nanoparticles (NO-np) were previously shown (see our Nanowerk Spotlight: "Nitric oxide releasing nanoparticles for effective treatment of infections") to accelerate wound healing in both uninfected and infected murine full thickness wound model. | |
| "Mechanistically, we found that NO-np treatment accelerates wound healing as a result of increasing tissue concentrations of TGF-b, a potent cytokine with broad and extensive effects on wound healing ranging from macrophage and fibroblast recruitment to collagen deposition to neoangiogenesis," explains Friedman. "Based on these findings, we hypothesized that the NO-np can accelerate healing in a murine model of impaired wound healing in the setting of immunodeficiency." | |
| In their new study, the researchers compare the efficacy of NO-np and DETA NONOate in non-obese, diabetic, severe combined immunodeficiency (NOD-SCID) mice. These mice may be used as a model of impaired wound healing due to immune compromise, as they lack T and B cells. | |
| Friedman points out that, similarly to previous results with the NO-np, it was found that treatment with NO-np significantly increased wound closure clinically, correlating to the increased and well organized collagen deposition and new vessel formation see histologically. | |
| "In addition, those wounds treated with the DETA NONOate histologically demonstrated poorly organized granulation tissue, degenerated collagen, and increased inflammation even when compared with untreated animals, suggesting that more research is needed to fully elucidate the safety of this NO donor," he says. "Our work also highlights the importance of lymphocytes as a source of NO and their role in wound healing." | |
| This pre-clinical work is very promising as managing wounds in patients with either inherent or iatrogenic immunosuppression is an extraordinary clinical and financial challenge. Because NO boasts numerous biological properties, its use in this setting may allow physicians to address and amerliorate the broad range of impediments ranging from microbial colonization/infection to poor perfusion to retarded migration of epithelial cells. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
