| Posted: Aug 31, 2012 | |
The challenge of testing nanomaterial ecotoxicity in aquatic environments |
|
| (Nanowerk Spotlight) Aquatic ecotoxicity test methods, which are routinely applied to testing of nanomaterials, were originally developed for water soluble chemicals. Nanomaterials are fundamentally different from many 'conventional' chemicals as they often have limited or no solubility at all and are potentially released to the environment in a particulate form (e.g. carbon nanotubes). Only limited nano-specific guidance on ecotoxicity testing is currently available, for instance existing preliminary OECD (eco)toxicity test guidance to nanomaterials (pdf) by the OECD Working Party on Manufactured Nanomaterials. These guidelines exhibit a number of specific shortcomings mainly related to characterization, exposure preparation, quantification and monitoring concentrations, and dose-metrics. | |
| A new paper from scientists in Denmark, published in the August 16, 2012 online edition of Nanotoxicology ("The challenges of testing metal and metal oxide nanoparticles in algal bioassays: titanium dioxide and gold nanoparticles as case studies"), contributes to the progress in algae testing of nanomaterials. This work also aids in the development of additional guidance as it adds to the understanding of pros and cons of different techniques for biomass quantification. | |
| "The motivation for carrying out this research was partly related to large variations in algal growth inhibition test results for nanoparticles – both in our own lab but also comparing results reported in the literature," Nanna Hartmann, who carried out this work as part of her PhD project ("Ecotoxicity of engineered nanoparticles to freshwater organisms") at the Department of Environmental Engineering at Technical University of Denmark (DTU), tells Nanowerk. "One explanation could be that the behavior of nanoparticles changes with media properties, over time and due to interactions with organisms. At the same time the nanoparticles have an effect on the organisms. Hence, we wanted to understand better how biomass quantification methods were influenced by these dynamic interactions and try to identify the most optimal technique." | |
 |
|
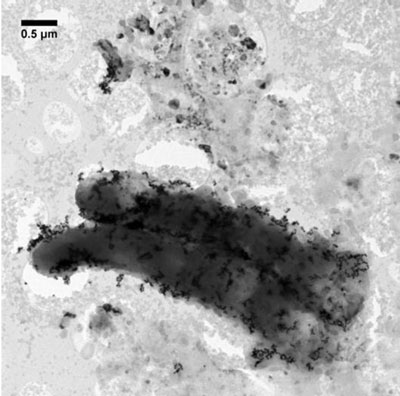
| Algal cells after 24 hours exposure to gold nanoparticles. (Image: Christian Engelbrekt, DTU) | |
| Although several papers exist on toxicity of nanomaterials to algae this is an issue which has previously been investigated and described in-depth (see for instance a paper on algal testing of titanium dioxide nanoparticles, also by the DTU team). | |
| If algal biomass can be correctly and accurately quantified, and the influence of artefacts eliminated, then the results of the algal growth inhibition tests will be much more reliable, comparable and applicable to risk assessment. | |
| To compare differences in algal toxicity of different nanoparticles a first step is to make sure that observed effects are actually toxic effects and not influenced by the biomass quantification technique. | |
| In their study, the team, led by Anders Baun, a professor at DTU who leads the Nanotechnology & Risk group and Professor Jens Ulstrup from DTU's Nanoscale Chemistry group, did an in-depth investigation of the method that is used for testing effects of chemicals on algal growth and its applicability to testing of nanomaterials toxicity. | |
| "Specifically, we evaluated three techniques which are routinely used for quantifying algal biomass, namely: coulter counting; cell counting by use of a haemocytometer; and measuring the fluorescence of algal pigment extracts," explains Hartmann. "Based on our results, we found that the fluorometric method was most suitable as the method allows for a physical separation of the nanoparticles and the biomass surrogate (pigment). This technique needs to be combined with visual inspections of the algal cells in a microscope. This provides additional information about the exposure of the cell, interactions between particles and cells, and it allows for detection of physiological effects." | |
| She notes that the fluorescence methods need some further optimization to minimize particle interference on the fluorescence measurements. | |
| The team's results also highlight another important issue: as the test system is dynamic, both quantitative and qualitative changes in exposure should be monitored over time. Otherwise time-depended changes in toxicity cannot retrospectively be linked to particle behavior and characteristics. | |
| The research team points out that there are still some challenges in the test designs that have to be solved. For example a better separation of particles and algal pigment extracts will decrease particle background interference. Also, there are other measuring techniques – such as e.g. flow cytometry – that could be evaluated and compared to the techniques investigated in this study. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
