| Posted: Sep 12, 2012 | |
Surface chemistry of silver nanoparticles and cell death |
|
| (Nanowerk Spotlight) Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae ("Surface defects on silver nanoparticles hold dangers for aquatic life"). Soil microorganisms can be affected when, for example, sewage sludge contaminated with nanosilver is spread on fields ("Researchers identify silver nanoparticles in sewage sludge of wastewater treatment plants") and nanosilver in waste water has therefore been identified by an international group of researchers as one of 15 areas of concern that can threaten biological diversity ("A horizon scan of global conservation issues for 2010"). Silver nanoparticles show negative effects on fishes and crustaceans already at low concentrations ("Silver nanoparticles can cause toxicity in fish"); in mammals, this material is toxic only at very high concentrations. | |
| Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. | |
| "Although there is some information about the toxicity of silver nanoparticles, the major part of the toxicity puzzle is still missing," Mustafa Culha, a professor who leads the Nanobiotechnology Group at Yeditepe University in Istanbul, Turkey, Tells Nanowerk. "For example, what is the influence of silver nanoparticles on cellular signaling pathways? Little is known and the missing pieces are needed to fill the gaps in our current understanding of how the cell response relates to the silver nanoparticle treatment as well as to cytotoxicity and genotoxicity." | |
| In a previous Nanowerk Spotlight ("Surface modification of silver nanoparticles and their interactions with living cells") we reported on work by Culha's team where they demonstrated that the functionalization of noble metal nanoparticles with multi-ligands can be used for cellular targeting. In this study, they also demonstrated that the toxicity of silver nanoparticles can be reduced by chemical modification, and they showed the possibility of using lactose-modified silver nanoparticles for selective targeting of eukaryotic cells. | |
| In their latest paper, reported in the August 24, 2012 online edition of Nanotechnology ("The influence of the surface chemistry of silver nanoparticles on cell death"), the researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles. | |
 |
|
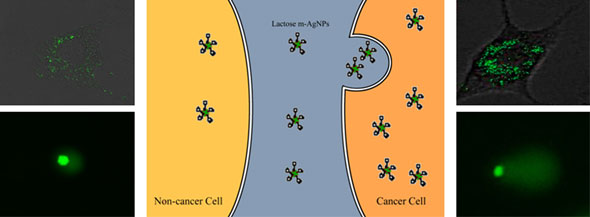
| A proposed model depicting cell-silver nanoparticle (AgNP) interactions. The lactose modified AgNPs entered into the cancer (A549) cells at a faster rate and triggered more cellular DNA damage, which led to upregulation of p53 gene inducing apoptosis in the cancer cells, compared to noncancer (HDF) cells. (Image: Dr. Culha, Yeditepe University) | |
| ""We demonstrate that the potential risks of silver nanoparticles to human health and the environment can be reduced by modifying their surface with a biological ligand," says Culha. "This reduces the cytotoxic, genotoxic, necrotic and apoptotic effects in healthy Human Dermal Fibroblast (HDF) cells. Additionally, our study adds to existing knowledge that modified silver nanoparticles play a protective role by causing different p53 gene related cell death effects in HDF and adenocarcinomic human alveolar basal epithelial A549 cells. Due to the different cellular responses, the modified silver nanoparticles might be used for the development of novel methods for selective killing of cancerous cells." | |
| In their work, the scientists found that lactose modified silver nanoparticles triggered more cellular DNA damage, which led to upregulation of the p53 gene inducing apoptosis in the A549 cells compared to HDF cells. These nanoparticles do not cause necrosis in the healthy HDF cells. Thus, the cancer cells can be killed by lactose modified silver nanoparticles by driving them to apoptosis while the surrounding healthy cells can be saved due to their lesser necrotic and apoptotic levels. | |
| The team specifically explored the influence of surface chemistry on the balance between toxicity and p53 gene mediated cell death in order to fill the missing pieces in this cell-silver nanoparticles interaction puzzle. In light of potential exposure and medical applications of silver nanoparticles, they chose the HDF and A549 cells to evaluate potential cell interaction of these silver nanoparticles. | |
| Culha notes that the HDF cells in the study were chosen because one of the major routes to exposure to nanoparticles is through the skin. A549 cells were chosen because there is limited information about the interaction between the nanoparticle-cancer cell interaction and toxicity. | |
| "Overall" he says, "our results provide more information about the impact of silver nanoparticles on human health and environment." | |
| For the A549 cells, the researchers observed the highest apoptosis and p53 gene expression with unmodified silver nanoparticles; this could therefore be a model nanoparticle for killing cancer cells using direct injection into the tumor. | |
| As Culha explains, the cancer cells can be killed with dual mechanisms by lactose modified silver nanoparticles via apoptosis and heating (photothermal therapy) while the surrounding healthy cells can be saved due to their diminished necrotic and apoptotic levels. | |
| "These results may help to develop novel therapies utilizing modified silver nanoparticles" he says. "However, the current level of understanding of the interaction between nanomaterials and living systems is not enough to make general statements and we need a bigger effort by researchers around the world to clarify these." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
